| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Steroid 17-alpha-hydroxylase/17,20 lyase |
|---|
| Ligand | BDBM50075391 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1473765 (CHEMBL3418490) |
|---|
| Ki | 1.9±n/a nM |
|---|
| Citation |  Njar, VC; Brodie, AM Discovery and development of Galeterone (TOK-001 or VN/124-1) for the treatment of all stages of prostate cancer. J Med Chem58:2077-87 (2015) [PubMed] Article Njar, VC; Brodie, AM Discovery and development of Galeterone (TOK-001 or VN/124-1) for the treatment of all stages of prostate cancer. J Med Chem58:2077-87 (2015) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Steroid 17-alpha-hydroxylase/17,20 lyase |
|---|
| Name: | Steroid 17-alpha-hydroxylase/17,20 lyase |
|---|
| Synonyms: | CP17A_HUMAN | CYP17 | CYP17A1 | CYPXVII | Cytochrome P450 17A1 | Cytochrome P450 C17 (CYP17 ) | Cytochrome P450 C17 (CYP17) | Cytochrome P450 CYP17 | Cytochrome P450-C17 | Cytochrome P450-C17 (CYP17) | P450-C17 | S17AH | Steroid 17-alpha-Monooxygenase (CYP17) | Steroid 17-alpha-hydroxylase/17,20 lyase | Steroid 17-alpha-monooxygenase | cytochrome P450 monooxygenase 17 alpha hydroxylase/17,20-lyase (CYP17) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57382.42 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | E.coli expressing human CYP17 |
|---|
| Residue: | 508 |
|---|
| Sequence: | MWELVALLLLTLAYLFWPKRRCPGAKYPKSLLSLPLVGSLPFLPRHGHMHNNFFKLQKKY
GPIYSVRMGTKTTVIVGHHQLAKEVLIKKGKDFSGRPQMATLDIASNNRKGIAFADSGAH
WQLHRRLAMATFALFKDGDQKLEKIICQEISTLCDMLATHNGQSIDISFPVFVAVTNVIS
LICFNTSYKNGDPELNVIQNYNEGIIDNLSKDSLVDLVPWLKIFPNKTLEKLKSHVKIRN
DLLNKILENYKEKFRSDSITNMLDTLMQAKMNSDNGNAGPDQDSELLSDNHILTTIGDIF
GAGVETTTSVVKWTLAFLLHNPQVKKKLYEEIDQNVGFSRTPTISDRNRLLLLEATIREV
LRLRPVAPMLIPHKANVDSSIGEFAVDKGTEVIINLWALHHNEKEWHQPDQFMPERFLNP
AGTQLISPSVSYLPFGAGPRSCIGEILARQELFLIMAWLLQRFDLEVPDDGQLPSLEGIP
KVVFLIDSFKVKIKVRQAWREAQAEGST
|
|
|
|---|
| BDBM50075391 |
|---|
| n/a |
|---|
| Name | BDBM50075391 |
|---|
| Synonyms: | CHEMBL3415124 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H28N2O |
|---|
| Mol. Mass. | 336.4705 |
|---|
| SMILES | [H][C@@]12CC=C(n3ccnc3)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])CCC2=CC(=O)CC[C@]12C |r,t:3,23| |
|---|
| Structure | 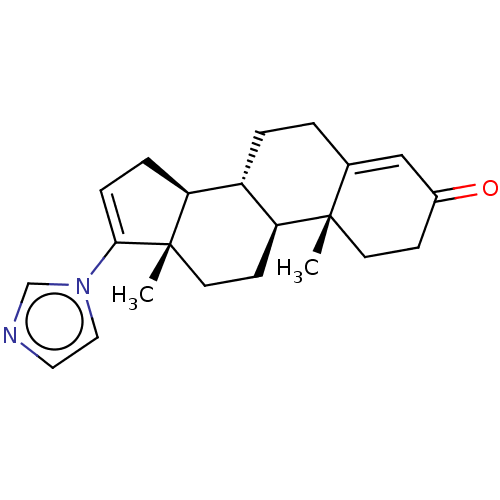
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Njar, VC; Brodie, AM Discovery and development of Galeterone (TOK-001 or VN/124-1) for the treatment of all stages of prostate cancer. J Med Chem58:2077-87 (2015) [PubMed] Article
Njar, VC; Brodie, AM Discovery and development of Galeterone (TOK-001 or VN/124-1) for the treatment of all stages of prostate cancer. J Med Chem58:2077-87 (2015) [PubMed] Article