| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Muscarinic acetylcholine receptor M3 |
|---|
| Ligand | BDBM50084425 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1477237 (CHEMBL3428929) |
|---|
| Ki | 0.630957±n/a nM |
|---|
| Citation |  Hughes, AD; Chen, Y; Hegde, SS; Jasper, JR; Jaw-Tsai, S; Lee, TW; McNamara, A; Pulido-Rios, MT; Steinfeld, T; Mammen, M Discovery of (R)-1-(3-((2-chloro-4-(((2-hydroxy-2-(8-hydroxy-2-oxo-1,2-dihydroquinolin-5-yl)ethyl)amino)methyl)-5-methoxyphenyl)amino)-3-oxopropyl)piperidin-4-yl [1,1'-biphenyl]-2-ylcarbamate (TD-5959, GSK961081, batefenterol): first-in-class dual pharmacology multivalent muscarinic antagonist.. J Med Chem58:2609-22 (2015) [PubMed] Article Hughes, AD; Chen, Y; Hegde, SS; Jasper, JR; Jaw-Tsai, S; Lee, TW; McNamara, A; Pulido-Rios, MT; Steinfeld, T; Mammen, M Discovery of (R)-1-(3-((2-chloro-4-(((2-hydroxy-2-(8-hydroxy-2-oxo-1,2-dihydroquinolin-5-yl)ethyl)amino)methyl)-5-methoxyphenyl)amino)-3-oxopropyl)piperidin-4-yl [1,1'-biphenyl]-2-ylcarbamate (TD-5959, GSK961081, batefenterol): first-in-class dual pharmacology multivalent muscarinic antagonist.. J Med Chem58:2609-22 (2015) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Muscarinic acetylcholine receptor M3 |
|---|
| Name: | Muscarinic acetylcholine receptor M3 |
|---|
| Synonyms: | ACM3_HUMAN | CHRM3 | Cholinergic, muscarinic M3 | Muscarinic Receptors M3 | Muscarinic receptor M3 | RecName: Full=Muscarinic acetylcholine receptor M3 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 66151.03 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P20309 |
|---|
| Residue: | 590 |
|---|
| Sequence: | MTLHNNSTTSPLFPNISSSWIHSPSDAGLPPGTVTHFGSYNVSRAAGNFSSPDGTTDDPL
GGHTVWQVVFIAFLTGILALVTIIGNILVIVSFKVNKQLKTVNNYFLLSLACADLIIGVI
SMNLFTTYIIMNRWALGNLACDLWLAIDYVASNASVMNLLVISFDRYFSITRPLTYRAKR
TTKRAGVMIGLAWVISFVLWAPAILFWQYFVGKRTVPPGECFIQFLSEPTITFGTAIAAF
YMPVTIMTILYWRIYKETEKRTKELAGLQASGTEAETENFVHPTGSSRSCSSYELQQQSM
KRSNRRKYGRCHFWFTTKSWKPSSEQMDQDHSSSDSWNNNDAAASLENSASSDEEDIGSE
TRAIYSIVLKLPGHSTILNSTKLPSSDNLQVPEEELGMVDLERKADKLQAQKSVDDGGSF
PKSFSKLPIQLESAVDTAKTSDVNSSVGKSTATLPLSFKEATLAKRFALKTRSQITKRKR
MSLVKEKKAAQTLSAILLAFIITWTPYNIMVLVNTFCDSCIPKTFWNLGYWLCYINSTVN
PVCYALCNKTFRTTFKMLLLCQCDKKKRRKQQYQQRQSVIFHKRAPEQAL
|
|
|
|---|
| BDBM50084425 |
|---|
| n/a |
|---|
| Name | BDBM50084425 |
|---|
| Synonyms: | CHEMBL3426704 | US9394275, I-25 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C41H45N5O7 |
|---|
| Mol. Mass. | 719.8253 |
|---|
| SMILES | COc1cc(ccc1CNC[C@H](O)c1ccc(O)c2[nH]c(=O)ccc12)N(C)C(=O)CCN1CCC(CC1)OC(=O)Nc1ccccc1-c1ccccc1 |r| |
|---|
| Structure | 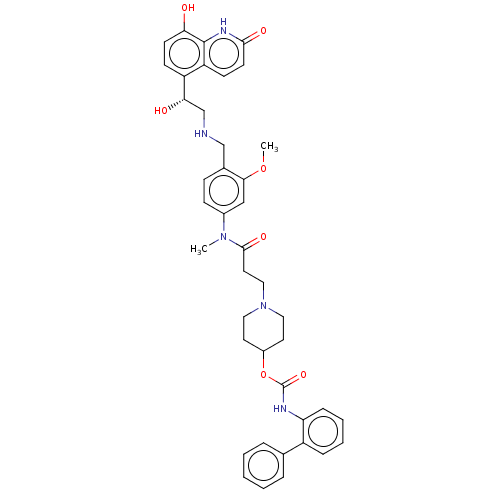
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Hughes, AD; Chen, Y; Hegde, SS; Jasper, JR; Jaw-Tsai, S; Lee, TW; McNamara, A; Pulido-Rios, MT; Steinfeld, T; Mammen, M Discovery of (R)-1-(3-((2-chloro-4-(((2-hydroxy-2-(8-hydroxy-2-oxo-1,2-dihydroquinolin-5-yl)ethyl)amino)methyl)-5-methoxyphenyl)amino)-3-oxopropyl)piperidin-4-yl [1,1'-biphenyl]-2-ylcarbamate (TD-5959, GSK961081, batefenterol): first-in-class dual pharmacology multivalent muscarinic antagonist.. J Med Chem58:2609-22 (2015) [PubMed] Article
Hughes, AD; Chen, Y; Hegde, SS; Jasper, JR; Jaw-Tsai, S; Lee, TW; McNamara, A; Pulido-Rios, MT; Steinfeld, T; Mammen, M Discovery of (R)-1-(3-((2-chloro-4-(((2-hydroxy-2-(8-hydroxy-2-oxo-1,2-dihydroquinolin-5-yl)ethyl)amino)methyl)-5-methoxyphenyl)amino)-3-oxopropyl)piperidin-4-yl [1,1'-biphenyl]-2-ylcarbamate (TD-5959, GSK961081, batefenterol): first-in-class dual pharmacology multivalent muscarinic antagonist.. J Med Chem58:2609-22 (2015) [PubMed] Article