| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50361983 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1488506 (CHEMBL3536348) |
|---|
| IC50 | 27±n/a nM |
|---|
| Citation |  Henne, KR; Tran, TB; VandenBrink, BM; Rock, DA; Aidasani, DK; Subramanian, R; Mason, AK; Stresser, DM; Teffera, Y; Wong, SG; Johnson, MG; Chen, X; Tonn, GR; Wong, BK Sequential metabolism of AMG 487, a novel CXCR3 antagonist, results in formation of quinone reactive metabolites that covalently modify CYP3A4 Cys239 and cause time-dependent inhibition of the enzyme. Drug Metab Dispos40:1429-40 (2012) [PubMed] Article Henne, KR; Tran, TB; VandenBrink, BM; Rock, DA; Aidasani, DK; Subramanian, R; Mason, AK; Stresser, DM; Teffera, Y; Wong, SG; Johnson, MG; Chen, X; Tonn, GR; Wong, BK Sequential metabolism of AMG 487, a novel CXCR3 antagonist, results in formation of quinone reactive metabolites that covalently modify CYP3A4 Cys239 and cause time-dependent inhibition of the enzyme. Drug Metab Dispos40:1429-40 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50361983 |
|---|
| n/a |
|---|
| Name | BDBM50361983 |
|---|
| Synonyms: | CHEMBL1939697 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C30H24F3N5O4 |
|---|
| Mol. Mass. | 575.5379 |
|---|
| SMILES | C[C@@H](N(Cc1cccnc1)C(=O)Cc1ccc(OC(F)(F)F)cc1)c1nc2ncccc2c(=O)n1-c1ccc(O)cc1 |r| |
|---|
| Structure | 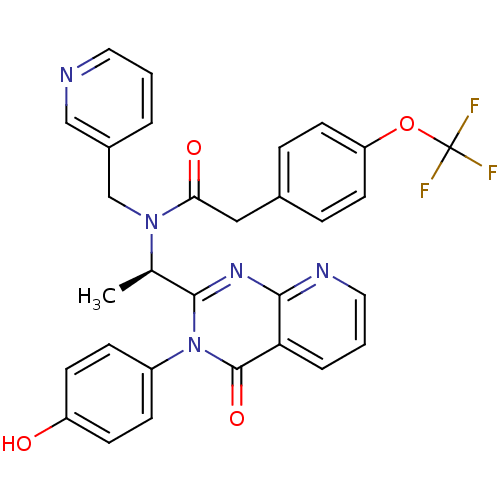
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Henne, KR; Tran, TB; VandenBrink, BM; Rock, DA; Aidasani, DK; Subramanian, R; Mason, AK; Stresser, DM; Teffera, Y; Wong, SG; Johnson, MG; Chen, X; Tonn, GR; Wong, BK Sequential metabolism of AMG 487, a novel CXCR3 antagonist, results in formation of quinone reactive metabolites that covalently modify CYP3A4 Cys239 and cause time-dependent inhibition of the enzyme. Drug Metab Dispos40:1429-40 (2012) [PubMed] Article
Henne, KR; Tran, TB; VandenBrink, BM; Rock, DA; Aidasani, DK; Subramanian, R; Mason, AK; Stresser, DM; Teffera, Y; Wong, SG; Johnson, MG; Chen, X; Tonn, GR; Wong, BK Sequential metabolism of AMG 487, a novel CXCR3 antagonist, results in formation of quinone reactive metabolites that covalently modify CYP3A4 Cys239 and cause time-dependent inhibition of the enzyme. Drug Metab Dispos40:1429-40 (2012) [PubMed] Article