| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Kappa-type opioid receptor |
|---|
| Ligand | BDBM50139013 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1498898 (CHEMBL3584105) |
|---|
| IC50 | >1000±n/a nM |
|---|
| Citation |  Piekielna, J; Perlikowska, R; do-Rego, JC; do-Rego, JL; Cerlesi, MC; Calo, G; Kluczyk, A; Lapinski, K; T�mb�ly, C; Janecka, A Synthesis of mixed opioid affinity cyclic endomorphin-2 analogues with fluorinated phenylalanines. ACS Med Chem Lett6:579-83 (2015) [PubMed] Article Piekielna, J; Perlikowska, R; do-Rego, JC; do-Rego, JL; Cerlesi, MC; Calo, G; Kluczyk, A; Lapinski, K; T�mb�ly, C; Janecka, A Synthesis of mixed opioid affinity cyclic endomorphin-2 analogues with fluorinated phenylalanines. ACS Med Chem Lett6:579-83 (2015) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Kappa-type opioid receptor |
|---|
| Name: | Kappa-type opioid receptor |
|---|
| Synonyms: | Kappa Opioid Receptor | OPIATE Kappa | OPRK1 | OPRK_CAVPO | Opiate Kappa 1 | mu/kappa opioid receptor |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 42744.99 |
|---|
| Organism: | Cavia porcellus (domestic guinea pig) |
|---|
| Description: | P41144 |
|---|
| Residue: | 380 |
|---|
| Sequence: | MGRRRQGPAQPASELPARNACLLPNGSAWLPGWAEPDGNGSAGPQDEQLEPAHISPAIPV
IITAVYSVVFVVGLVGNSLVMFVIIRYTKMKTATNIYIFNLALADALVTTTMPFQSTVYL
MNSWPFGDVLCKIVISIDYYNMFTSIFTLTMMSVDRYIAVCHPVKALDFRTPLKAKIINI
CIWLLSSSVGISAIILGGTKVREDVDIIECSLQFPDDDYSWWDLFMKICVFVFAFVIPVL
IIIVCYTLMILRLKSVRLLSGSREKDRNLRRITRLVLVVVAVFIICWTPIHIFILVEALG
STSHSTAALSSYYFCIALGYTNSSLNPILYAFLDENFKRCFRDFCFPIKMRMERQSTSRV
RNTVQDPAYMRNVDGVNKPV
|
|
|
|---|
| BDBM50139013 |
|---|
| n/a |
|---|
| Name | BDBM50139013 |
|---|
| Synonyms: | (S)-1-[(S)-2-Amino-3-(4-hydroxy-phenyl)-propionyl]-pyrrolidine-2-carboxylic acid [(S)-1-((S)-1-carbamoyl-2-phenyl-ethylcarbamoyl)-2-phenyl-ethyl]-amide | (S)-N-((S)-1-((S)-1-amino-1-oxo-3-phenylpropan-2-ylamino)-1-oxo-3-phenylpropan-2-yl)-1-((S)-2-amino-3-(4-hydroxyphenyl)propanoyl)pyrrolidine-2-carboxamide | 1-[(S)-2-Amino-3-(4-hydroxy-phenyl)-propionyl]-pyrrolidine-2-carboxylicacid[1-((S)-(S)-1-carbamoyl-2-phenyl-ethylcarbamoyl)-2-phenyl-ethyl]-amide | CHEMBL333357 | H-Tyr-Pro-Phe-Phe-NH2 | Tyr-Pro-Phe-Phe-NH2 | endomorphin-2 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C32H37N5O5 |
|---|
| Mol. Mass. | 571.6667 |
|---|
| SMILES | N[C@@H](Cc1ccc(O)cc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r| |
|---|
| Structure | 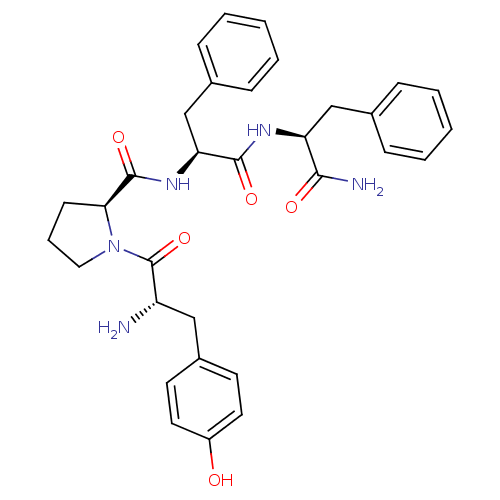
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Piekielna, J; Perlikowska, R; do-Rego, JC; do-Rego, JL; Cerlesi, MC; Calo, G; Kluczyk, A; Lapinski, K; T�mb�ly, C; Janecka, A Synthesis of mixed opioid affinity cyclic endomorphin-2 analogues with fluorinated phenylalanines. ACS Med Chem Lett6:579-83 (2015) [PubMed] Article
Piekielna, J; Perlikowska, R; do-Rego, JC; do-Rego, JL; Cerlesi, MC; Calo, G; Kluczyk, A; Lapinski, K; T�mb�ly, C; Janecka, A Synthesis of mixed opioid affinity cyclic endomorphin-2 analogues with fluorinated phenylalanines. ACS Med Chem Lett6:579-83 (2015) [PubMed] Article