| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Nitric oxide synthase, brain |
|---|
| Ligand | BDBM152712 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1500370 (CHEMBL3587003) |
|---|
| Ki | 17±n/a nM |
|---|
| Citation |  Kang, S; Li, H; Tang, W; Mart�sek, P; Roman, LJ; Poulos, TL; Silverman, RB 2-Aminopyridines with a Truncated Side Chain To Improve Human Neuronal Nitric Oxide Synthase Inhibitory Potency and Selectivity. J Med Chem58:5548-60 (2015) [PubMed] Article Kang, S; Li, H; Tang, W; Mart�sek, P; Roman, LJ; Poulos, TL; Silverman, RB 2-Aminopyridines with a Truncated Side Chain To Improve Human Neuronal Nitric Oxide Synthase Inhibitory Potency and Selectivity. J Med Chem58:5548-60 (2015) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Nitric oxide synthase, brain |
|---|
| Name: | Nitric oxide synthase, brain |
|---|
| Synonyms: | Bnos | N-NOS | NC-NOS | NOS | NOS type I nNOS | NOS1_RAT | Neuronal nitric oxide synthase | Neuronal nitric oxide synthase (nNOS) | Nitric Oxide Synthase, brain | Nitric oxide synthase (nNOS) | Nitric oxide synthase, brain (nNOS) | Nitric-oxide synthase, brain | Nitric-oxide synthase, brain (nNOS) | Nitrogen oxide synthase - neuronal | Nos1 | Peptidyl-cysteine S-nitrosylase NOS1 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 160570.98 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | Recombinant nNOS overexpressed in E. coli was used in enzyme assays. |
|---|
| Residue: | 1429 |
|---|
| Sequence: | MEENTFGVQQIQPNVISVRLFKRKVGGLGFLVKERVSKPPVIISDLIRGGAAEQSGLIQA
GDIILAVNDRPLVDLSYDSALEVLRGIASETHVVLILRGPEGFTTHLETTFTGDGTPKTI
RVTQPLGPPTKAVDLSHQPSASKDQSLAVDRVTGLGNGPQHAQGHGQGAGSVSQANGVAI
DPTMKSTKANLQDIGEHDELLKEIEPVLSILNSGSKATNRGGPAKAEMKDTGIQVDRDLD
GKSHKAPPLGGDNDRVFNDLWGKDNVPVILNNPYSEKEQSPTSGKQSPTKNGSPSRCPRF
LKVKNWETDVVLTDTLHLKSTLETGCTEHICMGSIMLPSQHTRKPEDVRTKDQLFPLAKE
FLDQYYSSIKRFGSKAHMDRLEEVNKEIESTSTYQLKDTELIYGAKHAWRNASRCVGRIQ
WSKLQVFDARDCTTAHGMFNYICNHVKYATNKGNLRSAITIFPQRTDGKHDFRVWNSQLI
RYAGYKQPDGSTLGDPANVQFTEICIQQGWKAPRGRFDVLPLLLQANGNDPELFQIPPEL
VLEVPIRHPKFDWFKDLGLKWYGLPAVSNMLLEIGGLEFSACPFSGWYMGTEIGVRDYCD
NSRYNILEEVAKKMDLDMRKTSSLWKDQALVEINIAVLYSFQSDKVTIVDHHSATESFIK
HMENEYRCRGGCPADWVWIVPPMSGSITPVFHQEMLNYRLTPSFEYQPDPWNTHVWKGTN
GTPTKRRAIGFKKLAEAVKFSAKLMGQAMAKRVKATILYATETGKSQAYAKTLCEIFKHA
FDAKAMSMEEYDIVHLEHEALVLVVTSTFGNGDPPENGEKFGCALMEMRHPNSVQEERKS
YKVRFNSVSSYSDSRKSSGDGPDLRDNFESTGPLANVRFSVFGLGSRAYPHFCAFGHAVD
TLLEELGGERILKMREGDELCGQEEAFRTWAKKVFKAACDVFCVGDDVNIEKPNNSLISN
DRSWKRNKFRLTYVAEAPDLTQGLSNVHKKRVSAARLLSRQNLQSPKFSRSTIFVRLHTN
GNQELQYQPGDHLGVFPGNHEDLVNALIERLEDAPPANHVVKVEMLEERNTALGVISNWK
DESRLPPCTIFQAFKYYLDITTPPTPLQLQQFASLATNEKEKQRLLVLSKGLQEYEEWKW
GKNPTMVEVLEEFPSIQMPATLLLTQLSLLQPRYYSISSSPDMYPDEVHLTVAIVSYHTR
DGEGPVHHGVCSSWLNRIQADDVVPCFVRGAPSFHLPRNPQVPCILVGPGTGIAPFRSFW
QQRQFDIQHKGMNPCPMVLVFGCRQSKIDHIYREETLQAKNKGVFRELYTAYSREPDRPK
KYVQDVLQEQLAESVYRALKEQGGHIYVCGDVTMAADVLKAIQRIMTQQGKLSEEDAGVF
ISRLRDDNRYHEDIFGVTLRTYEVTNRLRSESIAFIEESKKDADEVFSS
|
|
|
|---|
| BDBM152712 |
|---|
| n/a |
|---|
| Name | BDBM152712 |
|---|
| Synonyms: | 4-methyl-6-[2-(5-{methyl[2-(methylamino)ethyl]amino}pyridin-3-yl)ethyl]pyridin-2-amine (14) | US10759791, Compound 5 | US9951014, Name 14b | nNOS inhibitor, 2 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H25N5 |
|---|
| Mol. Mass. | 299.4139 |
|---|
| SMILES | CNCCN(C)c1cncc(CCc2cc(C)cc(N)n2)c1 |
|---|
| Structure | 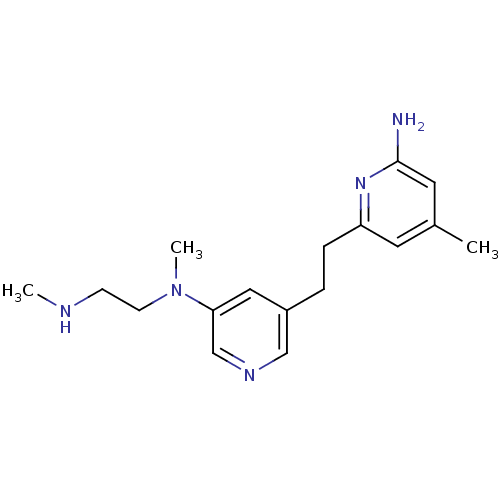
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kang, S; Li, H; Tang, W; Mart�sek, P; Roman, LJ; Poulos, TL; Silverman, RB 2-Aminopyridines with a Truncated Side Chain To Improve Human Neuronal Nitric Oxide Synthase Inhibitory Potency and Selectivity. J Med Chem58:5548-60 (2015) [PubMed] Article
Kang, S; Li, H; Tang, W; Mart�sek, P; Roman, LJ; Poulos, TL; Silverman, RB 2-Aminopyridines with a Truncated Side Chain To Improve Human Neuronal Nitric Oxide Synthase Inhibitory Potency and Selectivity. J Med Chem58:5548-60 (2015) [PubMed] Article