| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2J2 |
|---|
| Ligand | BDBM50103596 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1485701 (CHEMBL3541093) |
|---|
| IC50 | >50000±n/a nM |
|---|
| Citation |  Ren, S; Zeng, J; Mei, Y; Zhang, JZ; Yan, SF; Fei, J; Chen, L Discovery and characterization of novel, potent, and selective cytochrome P450 2J2 inhibitors. Drug Metab Dispos41:60-71 (2012) [PubMed] Article Ren, S; Zeng, J; Mei, Y; Zhang, JZ; Yan, SF; Fei, J; Chen, L Discovery and characterization of novel, potent, and selective cytochrome P450 2J2 inhibitors. Drug Metab Dispos41:60-71 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2J2 |
|---|
| Name: | Cytochrome P450 2J2 |
|---|
| Synonyms: | CP2J2_HUMAN | CYP2J2 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57623.04 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P51589 |
|---|
| Residue: | 502 |
|---|
| Sequence: | MLAAMGSLAAALWAVVHPRTLLLGTVAFLLAADFLKRRRPKNYPPGPWRLPFLGNFFLVD
FEQSHLEVQLFVKKYGNLFSLELGDISAVLITGLPLIKEALIHMDQNFGNRPVTPMREHI
FKKNGLIMSSGQAWKEQRRFTLTALRNFGLGKKSLEERIQEEAQHLTEAIKEENGQPFDP
HFKINNAVSNIICSITFGERFEYQDSWFQQLLKLLDEVTYLEASKTCQLYNVFPWIMKFL
PGPHQTLFSNWKKLKLFVSHMIDKHRKDWNPAETRDFIDAYLKEMSKHTGNPTSSFHEEN
LICSTLDLFFAGTETTSTTLRWALLYMALYPEIQEKVQAEIDRVIGQGQQPSTAARESMP
YTNAVIHEVQRMGNIIPLNVPREVTVDTTLAGYHLPKGTMILTNLTALHRDPTEWATPDT
FNPDHFLENGQFKKREAFMPFSIGKRACLGEQLARTELFIFFTSLMQKFTFRPPNNEKLS
LKFRMGITISPVSHRLCAVPQV
|
|
|
|---|
| BDBM50103596 |
|---|
| n/a |
|---|
| Name | BDBM50103596 |
|---|
| Synonyms: | (E)-2-hydroxy-5-((4-(N-pyridin-2-ylsulfamoyl)-phenyl)diazenyl) benzoic acid (A8) | Azulfidine | Azulfidine EN | Azulfidine EN-Tabs | CHEBI:9334 | S.A.S. | S.A.S.-500 | SAS-500 | Salazopyrin | Salazosulfapyridine | Salicylazosulfapyridine | Sulfasalazine | Sulphasalazine | US11744839, Compound of formula 1 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H14N4O5S |
|---|
| Mol. Mass. | 398.393 |
|---|
| SMILES | OC(=O)c1cc(ccc1O)\N=N\c1ccc(cc1)S(=O)(=O)Nc1ccccn1 |
|---|
| Structure | 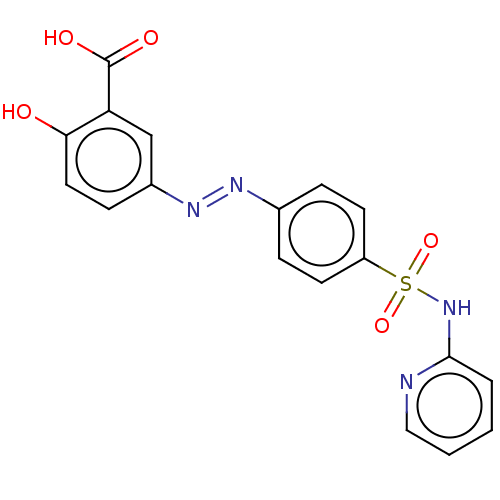
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ren, S; Zeng, J; Mei, Y; Zhang, JZ; Yan, SF; Fei, J; Chen, L Discovery and characterization of novel, potent, and selective cytochrome P450 2J2 inhibitors. Drug Metab Dispos41:60-71 (2012) [PubMed] Article
Ren, S; Zeng, J; Mei, Y; Zhang, JZ; Yan, SF; Fei, J; Chen, L Discovery and characterization of novel, potent, and selective cytochrome P450 2J2 inhibitors. Drug Metab Dispos41:60-71 (2012) [PubMed] Article