| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Glycogen synthase kinase-3 alpha |
|---|
| Ligand | BDBM50135286 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1546681 (CHEMBL3748206) |
|---|
| IC50 | >5000±n/a nM |
|---|
| Citation |  Reddy, MV; Akula, B; Jatiani, S; Vasquez-Del Carpio, R; Billa, VK; Mallireddigari, MR; Cosenza, SC; Venkata Subbaiah, DR; Bharathi, EV; Pallela, VR; Ramkumar, P; Jain, R; Aggarwal, AK; Reddy, EP Discovery of 2-(1H-indol-5-ylamino)-6-(2,4-difluorophenylsulfonyl)-8-methylpyrido[2,3-d]pyrimidin-7(8H)-one (7ao) as a potent selective inhibitor of Polo like kinase 2 (PLK2). Bioorg Med Chem24:521-44 (2016) [PubMed] Article Reddy, MV; Akula, B; Jatiani, S; Vasquez-Del Carpio, R; Billa, VK; Mallireddigari, MR; Cosenza, SC; Venkata Subbaiah, DR; Bharathi, EV; Pallela, VR; Ramkumar, P; Jain, R; Aggarwal, AK; Reddy, EP Discovery of 2-(1H-indol-5-ylamino)-6-(2,4-difluorophenylsulfonyl)-8-methylpyrido[2,3-d]pyrimidin-7(8H)-one (7ao) as a potent selective inhibitor of Polo like kinase 2 (PLK2). Bioorg Med Chem24:521-44 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Glycogen synthase kinase-3 alpha |
|---|
| Name: | Glycogen synthase kinase-3 alpha |
|---|
| Synonyms: | GSK-3 alpha | GSK3A | GSK3A_HUMAN | Glycogen synthase kinase 3 alpha (GSKalpha) | Glycogen synthase kinase-3 | Glycogen synthase kinase-3 alpha | Glycogen synthase kinase-3 alpha (GSK3 Alpha) | Glycogen synthase kinase-3 alpha (GSK3A) | Glycogen synthase kinase-3 alpha (GSK3alpha) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 50991.79 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P49840 |
|---|
| Residue: | 483 |
|---|
| Sequence: | MSGGGPSGGGPGGSGRARTSSFAEPGGGGGGGGGGPGGSASGPGGTGGGKASVGAMGGGV
GASSSGGGPGGSGGGGSGGPGAGTSFPPPGVKLGRDSGKVTTVVATLGQGPERSQEVAYT
DIKVIGNGSFGVVYQARLAETRELVAIKKVLQDKRFKNRELQIMRKLDHCNIVRLRYFFY
SSGEKKDELYLNLVLEYVPETVYRVARHFTKAKLTIPILYVKVYMYQLFRSLAYIHSQGV
CHRDIKPQNLLVDPDTAVLKLCDFGSAKQLVRGEPNVSYICSRYYRAPELIFGATDYTSS
IDVWSAGCVLAELLLGQPIFPGDSGVDQLVEIIKVLGTPTREQIREMNPNYTEFKFPQIK
AHPWTKVFKSRTPPEAIALCSSLLEYTPSSRLSPLEACAHSFFDELRCLGTQLPNNRPLP
PLFNFSAGELSIQPSLNAILIPPHLRSPAGTTTLTPSSQALTETPTSSDWQSTDATPTLT
NSS
|
|
|
|---|
| BDBM50135286 |
|---|
| n/a |
|---|
| Name | BDBM50135286 |
|---|
| Synonyms: | CHEMBL3745885 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H15F2N5O3S |
|---|
| Mol. Mass. | 467.448 |
|---|
| SMILES | Cn1c2nc(Nc3ccc4[nH]ccc4c3)ncc2cc(c1=O)S(=O)(=O)c1ccc(F)cc1F |
|---|
| Structure | 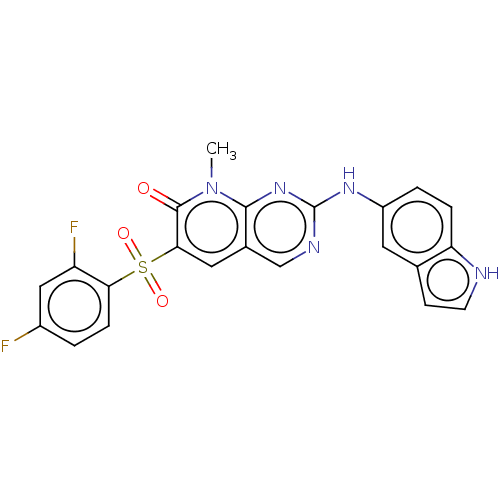
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Reddy, MV; Akula, B; Jatiani, S; Vasquez-Del Carpio, R; Billa, VK; Mallireddigari, MR; Cosenza, SC; Venkata Subbaiah, DR; Bharathi, EV; Pallela, VR; Ramkumar, P; Jain, R; Aggarwal, AK; Reddy, EP Discovery of 2-(1H-indol-5-ylamino)-6-(2,4-difluorophenylsulfonyl)-8-methylpyrido[2,3-d]pyrimidin-7(8H)-one (7ao) as a potent selective inhibitor of Polo like kinase 2 (PLK2). Bioorg Med Chem24:521-44 (2016) [PubMed] Article
Reddy, MV; Akula, B; Jatiani, S; Vasquez-Del Carpio, R; Billa, VK; Mallireddigari, MR; Cosenza, SC; Venkata Subbaiah, DR; Bharathi, EV; Pallela, VR; Ramkumar, P; Jain, R; Aggarwal, AK; Reddy, EP Discovery of 2-(1H-indol-5-ylamino)-6-(2,4-difluorophenylsulfonyl)-8-methylpyrido[2,3-d]pyrimidin-7(8H)-one (7ao) as a potent selective inhibitor of Polo like kinase 2 (PLK2). Bioorg Med Chem24:521-44 (2016) [PubMed] Article