| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Neuronal acetylcholine receptor subunit alpha-7 |
|---|
| Ligand | BDBM50143314 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1552367 (CHEMBL3761531) |
|---|
| Ki | 22±n/a nM |
|---|
| Citation |  Matera, C; Quadri, M; Sciaccaluga, M; Pom�, DY; Fasoli, F; De Amici, M; Fucile, S; Gotti, C; Dallanoce, C; Grazioso, G Modification of the anabaseine pyridine nucleus allows achieving binding and functional selectivity for thea3�4 nicotinic acetylcholine receptor subtype. Eur J Med Chem108:392-405 (2016) [PubMed] Article Matera, C; Quadri, M; Sciaccaluga, M; Pom�, DY; Fasoli, F; De Amici, M; Fucile, S; Gotti, C; Dallanoce, C; Grazioso, G Modification of the anabaseine pyridine nucleus allows achieving binding and functional selectivity for thea3�4 nicotinic acetylcholine receptor subtype. Eur J Med Chem108:392-405 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Neuronal acetylcholine receptor subunit alpha-7 |
|---|
| Name: | Neuronal acetylcholine receptor subunit alpha-7 |
|---|
| Synonyms: | ACHA7_HUMAN | CHRNA7 | Cholinergic, Nicotinic Alpha7 | NACHRA7 | Neuronal acetylcholine receptor protein alpha-7 subunit | Neuronal acetylcholine receptor subunit alpha-7 (nAChR-alpha 7) | Nicotinic acetylcholine receptor alpha-7 (alpha7 nAChR) |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | 56448.33 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | CHRNA7 (NACHRA7) |
|---|
| Residue: | 502 |
|---|
| Sequence: | MRCSPGGVWLALAASLLHVSLQGEFQRKLYKELVKNYNPLERPVANDSQPLTVYFSLSLL
QIMDVDEKNQVLTTNIWLQMSWTDHYLQWNVSEYPGVKTVRFPDGQIWKPDILLYNSADE
RFDATFHTNVLVNSSGHCQYLPPGIFKSSCYIDVRWFPFDVQHCKLKFGSWSYGGWSLDL
QMQEADISGYIPNGEWDLVGIPGKRSERFYECCKEPYPDVTFTVTMRRRTLYYGLNLLIP
CVLISALALLVFLLPADSGEKISLGITVLLSLTVFMLLVAEIMPATSDSVPLIAQYFAST
MIIVGLSVVVTVIVLQYHHHDPDGGKMPKWTRVILLNWCAWFLRMKRPGEDKVRPACQHK
QRRCSLASVEMSAVAPPPASNGNLLYIGFRGLDGVHCVPTPDSGVVCGRMACSPTHDEHL
LHGGQPPEGDPDLAKILEEVRYIANRFRCQDESEAVCSEWKFAACVVDRLCLMAFSVFTI
ICTIGILMSAPNFVEAVSKDFA
|
|
|
|---|
| BDBM50143314 |
|---|
| n/a |
|---|
| Name | BDBM50143314 |
|---|
| Synonyms: | (+)-Epibatidine | (-)-epibatidine | (1R,2R,4S)-2-(6-Chloro-pyridin-3-yl)-7-aza-bicyclo[2.2.1]heptane | 2-(6-Chloro-pyridin-3-yl)-7-aza-bicyclo[2.2.1]heptane (epibatidine) | CHEMBL298826 | EPIBATIDINE |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C11H13ClN2 |
|---|
| Mol. Mass. | 208.687 |
|---|
| SMILES | Clc1ccc(cn1)[C@H]1C[C@@H]2CC[C@H]1N2 |THB:4:7:13:11.10| |
|---|
| Structure | 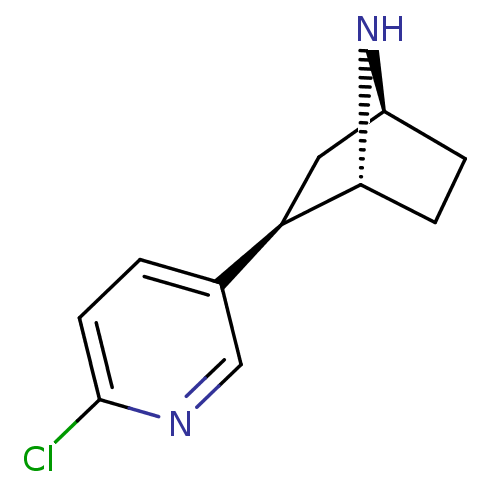
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Matera, C; Quadri, M; Sciaccaluga, M; Pom�, DY; Fasoli, F; De Amici, M; Fucile, S; Gotti, C; Dallanoce, C; Grazioso, G Modification of the anabaseine pyridine nucleus allows achieving binding and functional selectivity for thea3�4 nicotinic acetylcholine receptor subtype. Eur J Med Chem108:392-405 (2016) [PubMed] Article
Matera, C; Quadri, M; Sciaccaluga, M; Pom�, DY; Fasoli, F; De Amici, M; Fucile, S; Gotti, C; Dallanoce, C; Grazioso, G Modification of the anabaseine pyridine nucleus allows achieving binding and functional selectivity for thea3�4 nicotinic acetylcholine receptor subtype. Eur J Med Chem108:392-405 (2016) [PubMed] Article