| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Indoleamine 2,3-dioxygenase 1 |
|---|
| Ligand | BDBM50146460 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1555056 (CHEMBL3767634) |
|---|
| IC50 | 82±n/a nM |
|---|
| Citation |  Malachowski, WP; Winters, M; DuHadaway, JB; Lewis-Ballester, A; Badir, S; Wai, J; Rahman, M; Sheikh, E; LaLonde, JM; Yeh, SR; Prendergast, GC; Muller, AJ O-alkylhydroxylamines as rationally-designed mechanism-based inhibitors of indoleamine 2,3-dioxygenase-1. Eur J Med Chem108:564-76 (2016) [PubMed] Article Malachowski, WP; Winters, M; DuHadaway, JB; Lewis-Ballester, A; Badir, S; Wai, J; Rahman, M; Sheikh, E; LaLonde, JM; Yeh, SR; Prendergast, GC; Muller, AJ O-alkylhydroxylamines as rationally-designed mechanism-based inhibitors of indoleamine 2,3-dioxygenase-1. Eur J Med Chem108:564-76 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Indoleamine 2,3-dioxygenase 1 |
|---|
| Name: | Indoleamine 2,3-dioxygenase 1 |
|---|
| Synonyms: | I23O1_HUMAN | IDO | IDO-1 | IDO1 | INDO | Indoleamine 2,3-Dioxygenasae (IDO) | Indoleamine 2,3-dioxygenase | Indoleamine-pyrrole 2,3-dioxygenase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 45330.80 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P14902 |
|---|
| Residue: | 403 |
|---|
| Sequence: | MAHAMENSWTISKEYHIDEEVGFALPNPQENLPDFYNDWMFIAKHLPDLIESGQLRERVE
KLNMLSIDHLTDHKSQRLARLVLGCITMAYVWGKGHGDVRKVLPRNIAVPYCQLSKKLEL
PPILVYADCVLANWKKKDPNKPLTYENMDVLFSFRDGDCSKGFFLVSLLVEIAAASAIKV
IPTVFKAMQMQERDTLLKALLEIASCLEKALQVFHQIHDHVNPKAFFSVLRIYLSGWKGN
PQLSDGLVYEGFWEDPKEFAGGSAGQSSVFQCFDVLLGIQQTAGGGHAAQFLQDMRRYMP
PAHRNFLCSLESNPSVREFVLSKGDAGLREAYDACVKALVSLRSYHLQIVTKYILIPASQ
QPKENKTSEDPSKLEAKGTGGTDLMNFLKTVRSTTEKSLLKEG
|
|
|
|---|
| BDBM50146460 |
|---|
| n/a |
|---|
| Name | BDBM50146460 |
|---|
| Synonyms: | CHEMBL3763688 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C7H9ClINO |
|---|
| Mol. Mass. | 285.51 |
|---|
| SMILES | Cl.NOCc1ccc(I)cc1 |
|---|
| Structure | 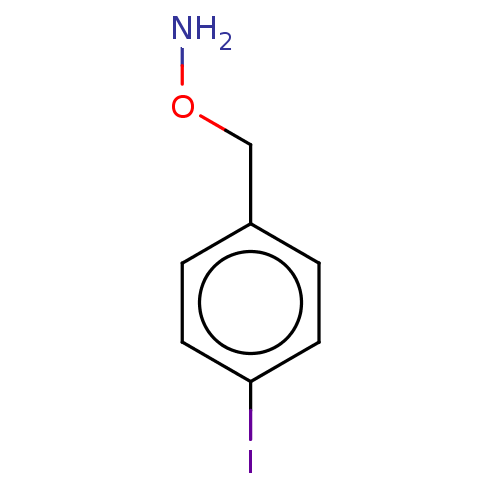
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Malachowski, WP; Winters, M; DuHadaway, JB; Lewis-Ballester, A; Badir, S; Wai, J; Rahman, M; Sheikh, E; LaLonde, JM; Yeh, SR; Prendergast, GC; Muller, AJ O-alkylhydroxylamines as rationally-designed mechanism-based inhibitors of indoleamine 2,3-dioxygenase-1. Eur J Med Chem108:564-76 (2016) [PubMed] Article
Malachowski, WP; Winters, M; DuHadaway, JB; Lewis-Ballester, A; Badir, S; Wai, J; Rahman, M; Sheikh, E; LaLonde, JM; Yeh, SR; Prendergast, GC; Muller, AJ O-alkylhydroxylamines as rationally-designed mechanism-based inhibitors of indoleamine 2,3-dioxygenase-1. Eur J Med Chem108:564-76 (2016) [PubMed] Article