| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Adenosine receptor A3 |
|---|
| Ligand | BDBM50118812 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1560589 (CHEMBL3777321) |
|---|
| Ki | 1.8±n/a nM |
|---|
| Citation |  Dosa, PI; Amin, EA Tactical Approaches to Interconverting GPCR Agonists and Antagonists. J Med Chem59:810-40 (2016) [PubMed] Article Dosa, PI; Amin, EA Tactical Approaches to Interconverting GPCR Agonists and Antagonists. J Med Chem59:810-40 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Adenosine receptor A3 |
|---|
| Name: | Adenosine receptor A3 |
|---|
| Synonyms: | A3 adenosine receptor (hA3) | AA3R_HUMAN | ADORA3 | Adenosine A3 receptor (A3AR) |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 36197.32 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P0DMS8 |
|---|
| Residue: | 318 |
|---|
| Sequence: | MPNNSTALSLANVTYITMEIFIGLCAIVGNVLVICVVKLNPSLQTTTFYFIVSLALADIA
VGVLVMPLAIVVSLGITIHFYSCLFMTCLLLIFTHASIMSLLAIAVDRYLRVKLTVRYKR
VTTHRRIWLALGLCWLVSFLVGLTPMFGWNMKLTSEYHRNVTFLSCQFVSVMRMDYMVYF
SFLTWIFIPLVVMCAIYLDIFYIIRNKLSLNLSNSKETGAFYGREFKTAKSLFLVLFLFA
LSWLPLSIINCIIYFNGEVPQLVLYMGILLSHANSMMNPIVYAYKIKKFKETYLLILKAC
VVCHPSDSLDTSIEKNSE
|
|
|
|---|
| BDBM50118812 |
|---|
| n/a |
|---|
| Name | BDBM50118812 |
|---|
| Synonyms: | (2S,3S,4R,5R)-3,4-Dihydroxy-5-[6-(3-iodo-benzylamino)-purin-9-yl]-tetrahydro-furan-2-carboxylic acid methylamide | (2S,3S,4R,5R)-3,4-dihydroxy-5-(6-(3-iodobenzylamino)-9H-purin-9-yl)-N-methyltetrahydrofuran-2-carboxamide | (2S,3S,4R,5R)-5-(6-(3-iodobenzylamino)-9H-purin-9-yl)-3,4-dihydroxy-N-methyl-tetrahydrofuran-2-carboxamide | (3R,4S,5R)-3,4-Dihydroxy-5-[6-(3-iodo-benzylamino)-purin-9-yl]-tetrahydro-furan-2-carboxylic acid methylamide | (3S,4R,5R)-3,4-Dihydroxy-5-[6-(3-iodo-benzylamino)-purin-9-yl]-tetrahydro-furan-2-carboxylic acid methylamide | (N-6-(3-iodobenzyl)adenosine-5'-N-methyluronamide | 3,4-Dihydroxy-5-[6-(3-iodo-benzylamino)-purin-9-yl]-tetrahydro-furan-2-carboxylic acid methylamide | CHEMBL119709 | IB-MECA | N6-(3-iodobenzyl)-5'-N-methylcarboxamidoadenosine |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H19IN6O4 |
|---|
| Mol. Mass. | 510.2857 |
|---|
| SMILES | CNC(=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(NCc3cccc(I)c3)ncnc12 |r| |
|---|
| Structure | 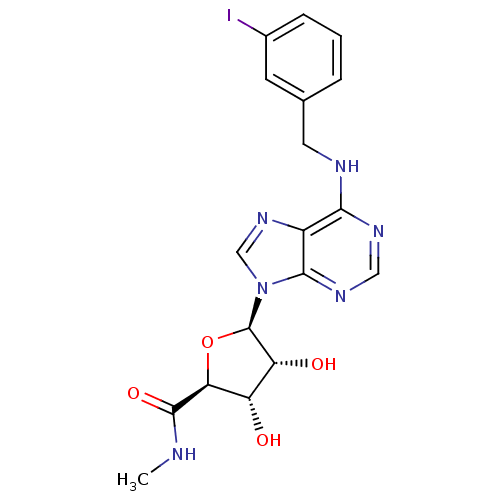
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Dosa, PI; Amin, EA Tactical Approaches to Interconverting GPCR Agonists and Antagonists. J Med Chem59:810-40 (2016) [PubMed] Article
Dosa, PI; Amin, EA Tactical Approaches to Interconverting GPCR Agonists and Antagonists. J Med Chem59:810-40 (2016) [PubMed] Article