| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cannabinoid receptor 2 |
|---|
| Ligand | BDBM50180040 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1565590 (CHEMBL3782259) |
|---|
| Ki | 4.7±n/a nM |
|---|
| Citation |  Mugnaini, C; Brizzi, A; Ligresti, A; Allar�, M; Lamponi, S; Vacondio, F; Silva, C; Mor, M; Di Marzo, V; Corelli, F Investigations on the 4-Quinolone-3-carboxylic Acid Motif. 7. Synthesis and Pharmacological Evaluation of 4-Quinolone-3-carboxamides and 4-Hydroxy-2-quinolone-3-carboxamides as High Affinity Cannabinoid Receptor 2 (CB2R) Ligands with Improved Aqueous Solubility. J Med Chem59:1052-67 (2016) [PubMed] Article Mugnaini, C; Brizzi, A; Ligresti, A; Allar�, M; Lamponi, S; Vacondio, F; Silva, C; Mor, M; Di Marzo, V; Corelli, F Investigations on the 4-Quinolone-3-carboxylic Acid Motif. 7. Synthesis and Pharmacological Evaluation of 4-Quinolone-3-carboxamides and 4-Hydroxy-2-quinolone-3-carboxamides as High Affinity Cannabinoid Receptor 2 (CB2R) Ligands with Improved Aqueous Solubility. J Med Chem59:1052-67 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cannabinoid receptor 2 |
|---|
| Name: | Cannabinoid receptor 2 |
|---|
| Synonyms: | CANNABINOID CB2 | CB-2 | CB2 | CB2A | CB2B | CNR2 | CNR2_HUMAN | CX5 | Cannabinoid CB2 receptor | Cannabinoid receptor 2 (CB2) | Cannabinoid receptor 2 (CB2R) | hCB2 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 39690.94 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P34972 |
|---|
| Residue: | 360 |
|---|
| Sequence: | MEECWVTEIANGSKDGLDSNPMKDYMILSGPQKTAVAVLCTLLGLLSALENVAVLYLILS
SHQLRRKPSYLFIGSLAGADFLASVVFACSFVNFHVFHGVDSKAVFLLKIGSVTMTFTAS
VGSLLLTAIDRYLCLRYPPSYKALLTRGRALVTLGIMWVLSALVSYLPLMGWTCCPRPCS
ELFPLIPNDYLLSWLLFIAFLFSGIIYTYGHVLWKAHQHVASLSGHQDRQVPGMARMRLD
VRLAKTLGLVLAVLLICWFPVLALMAHSLATTLSDQVKKAFAFCSMLCLINSMVNPVIYA
LRSGEIRSSAHHCLAHWKKCVRGLGSEAKEEAPRSSVTETEADGKITPWPDSRDLDLSDC
|
|
|
|---|
| BDBM50180040 |
|---|
| n/a |
|---|
| Name | BDBM50180040 |
|---|
| Synonyms: | 4-Oxo-1-pentyl-1,4-dihydro-quinoline-3-carboxylic acid adamantan-1-ylamide | CHEMBL201571 | N3-(1-adamantyl)-4-oxo-1-pentyl-1,4-dihydroquinoline-3-carboxamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H32N2O2 |
|---|
| Mol. Mass. | 392.5338 |
|---|
| SMILES | CCCCCn1cc(C(=O)NC23CC4CC(CC(C4)C2)C3)c(=O)c2ccccc12 |TLB:10:11:14:18.16.17,THB:16:15:12:18.17.19,16:17:14.15.20:12,19:17:14:20.11.12,19:11:14:18.16.17| |
|---|
| Structure | 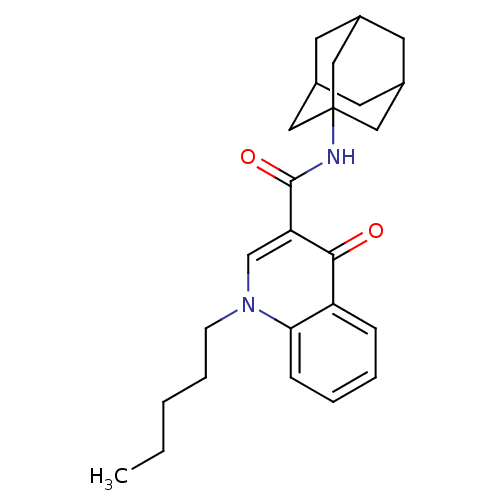
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Mugnaini, C; Brizzi, A; Ligresti, A; Allar�, M; Lamponi, S; Vacondio, F; Silva, C; Mor, M; Di Marzo, V; Corelli, F Investigations on the 4-Quinolone-3-carboxylic Acid Motif. 7. Synthesis and Pharmacological Evaluation of 4-Quinolone-3-carboxamides and 4-Hydroxy-2-quinolone-3-carboxamides as High Affinity Cannabinoid Receptor 2 (CB2R) Ligands with Improved Aqueous Solubility. J Med Chem59:1052-67 (2016) [PubMed] Article
Mugnaini, C; Brizzi, A; Ligresti, A; Allar�, M; Lamponi, S; Vacondio, F; Silva, C; Mor, M; Di Marzo, V; Corelli, F Investigations on the 4-Quinolone-3-carboxylic Acid Motif. 7. Synthesis and Pharmacological Evaluation of 4-Quinolone-3-carboxamides and 4-Hydroxy-2-quinolone-3-carboxamides as High Affinity Cannabinoid Receptor 2 (CB2R) Ligands with Improved Aqueous Solubility. J Med Chem59:1052-67 (2016) [PubMed] Article