

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Cytochrome P450 1A2 | ||
| Ligand | BDBM50167285 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_1573782 (CHEMBL3803203) | ||
| IC50 | <50119±n/a nM | ||
| Citation |  McBride, C; Cheruvallath, Z; Komandla, M; Tang, M; Farrell, P; Lawson, JD; Vanderpool, D; Wu, Y; Dougan, DR; Plonowski, A; Holub, C; Larson, C Discovery of potent, reversible MetAP2 inhibitors via fragment based drug discovery and structure based drug design-Part 2. Bioorg Med Chem Lett26:2779-2783 (2016) [PubMed] Article McBride, C; Cheruvallath, Z; Komandla, M; Tang, M; Farrell, P; Lawson, JD; Vanderpool, D; Wu, Y; Dougan, DR; Plonowski, A; Holub, C; Larson, C Discovery of potent, reversible MetAP2 inhibitors via fragment based drug discovery and structure based drug design-Part 2. Bioorg Med Chem Lett26:2779-2783 (2016) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Cytochrome P450 1A2 | |||
| Name: | Cytochrome P450 1A2 | ||
| Synonyms: | CP1A2_HUMAN | CYP1A2 | CYPIA2 | Cholesterol 25-hydroxylase | Cytochrome P(3)450 | Cytochrome P450 1A | Cytochrome P450 1A2 (CYP1A2) | Cytochrome P450 4 | Cytochrome P450-P3 | ||
| Type: | Enzyme | ||
| Mol. Mass.: | 58423.38 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P05177 | ||
| Residue: | 516 | ||
| Sequence: |
| ||
| BDBM50167285 | |||
| n/a | |||
| Name | BDBM50167285 | ||
| Synonyms: | CHEMBL3797780 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C16H11FN6 | ||
| Mol. Mass. | 306.2971 | ||
| SMILES | Cc1c(cnc2[nH]cnc12)-n1c2cn[nH]c2c2cc(F)ccc12 |(-2.83,-3.91,;-1.7,-4.41,;-.44,-3.51,;.97,-4.14,;1.13,-5.69,;-.13,-6.58,;-.31,-8.1,;-1.82,-8.4,;-2.56,-7.08,;-1.53,-5.95,;-.45,-1.97,;.76,-1.05,;2.33,-1.05,;2.8,.38,;1.54,1.31,;.31,.38,;-1.24,.38,;-2.28,1.57,;-3.78,1.21,;-4.58,2.15,;-4.28,,;-3.23,-1.4,;-1.71,-1.05,)| | ||
| Structure | 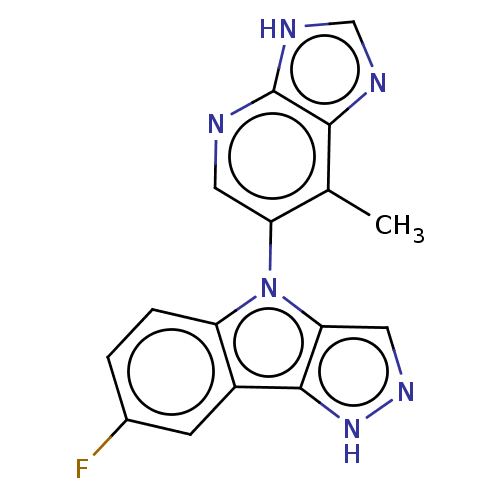
| ||