| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 1A2 |
|---|
| Ligand | BDBM50167916 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1574299 (CHEMBL3802684) |
|---|
| IC50 | 316±n/a nM |
|---|
| Citation |  Cheruvallath, Z; Tang, M; McBride, C; Komandla, M; Miura, J; Ton-Nu, T; Erikson, P; Feng, J; Farrell, P; Lawson, JD; Vanderpool, D; Wu, Y; Dougan, DR; Plonowski, A; Holub, C; Larson, C Discovery of potent, reversible MetAP2 inhibitors via fragment based drug discovery and structure based drug design-Part 1. Bioorg Med Chem Lett26:2774-2778 (2016) [PubMed] Article Cheruvallath, Z; Tang, M; McBride, C; Komandla, M; Miura, J; Ton-Nu, T; Erikson, P; Feng, J; Farrell, P; Lawson, JD; Vanderpool, D; Wu, Y; Dougan, DR; Plonowski, A; Holub, C; Larson, C Discovery of potent, reversible MetAP2 inhibitors via fragment based drug discovery and structure based drug design-Part 1. Bioorg Med Chem Lett26:2774-2778 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 1A2 |
|---|
| Name: | Cytochrome P450 1A2 |
|---|
| Synonyms: | CP1A2_HUMAN | CYP1A2 | CYPIA2 | Cholesterol 25-hydroxylase | Cytochrome P(3)450 | Cytochrome P450 1A | Cytochrome P450 1A2 (CYP1A2) | Cytochrome P450 4 | Cytochrome P450-P3 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 58423.38 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P05177 |
|---|
| Residue: | 516 |
|---|
| Sequence: | MALSQSVPFSATELLLASAIFCLVFWVLKGLRPRVPKGLKSPPEPWGWPLLGHVLTLGKN
PHLALSRMSQRYGDVLQIRIGSTPVLVLSRLDTIRQALVRQGDDFKGRPDLYTSTLITDG
QSLTFSTDSGPVWAARRRLAQNALNTFSIASDPASSSSCYLEEHVSKEAKALISRLQELM
AGPGHFDPYNQVVVSVANVIGAMCFGQHFPESSDEMLSLVKNTHEFVETASSGNPLDFFP
ILRYLPNPALQRFKAFNQRFLWFLQKTVQEHYQDFDKNSVRDITGALFKHSKKGPRASGN
LIPQEKIVNLVNDIFGAGFDTVTTAISWSLMYLVTKPEIQRKIQKELDTVIGRERRPRLS
DRPQLPYLEAFILETFRHSSFLPFTIPHSTTRDTTLNGFYIPKKCCVFVNQWQVNHDPEL
WEDPSEFRPERFLTADGTAINKPLSEKMMLFGMGKRRCIGEVLAKWEIFLFLAILLQQLE
FSVPPGVKVDLTPIYGLTMKHARCEHVQARLRFSIN
|
|
|
|---|
| BDBM50167916 |
|---|
| n/a |
|---|
| Name | BDBM50167916 |
|---|
| Synonyms: | CHEMBL3798615 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C14H7F3N4 |
|---|
| Mol. Mass. | 288.2274 |
|---|
| SMILES | FC(F)(F)c1cc(-c2cncc(c2)C#N)c2cn[nH]c2c1 |
|---|
| Structure | 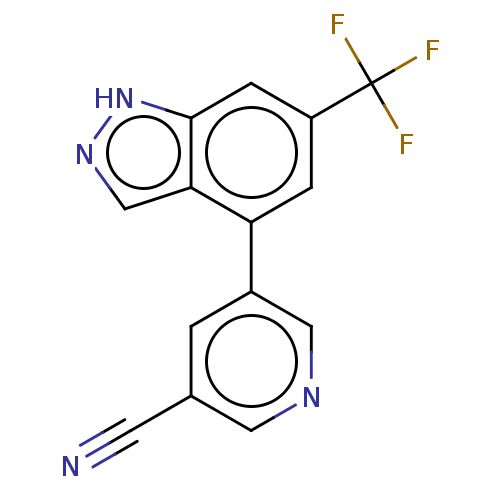
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Cheruvallath, Z; Tang, M; McBride, C; Komandla, M; Miura, J; Ton-Nu, T; Erikson, P; Feng, J; Farrell, P; Lawson, JD; Vanderpool, D; Wu, Y; Dougan, DR; Plonowski, A; Holub, C; Larson, C Discovery of potent, reversible MetAP2 inhibitors via fragment based drug discovery and structure based drug design-Part 1. Bioorg Med Chem Lett26:2774-2778 (2016) [PubMed] Article
Cheruvallath, Z; Tang, M; McBride, C; Komandla, M; Miura, J; Ton-Nu, T; Erikson, P; Feng, J; Farrell, P; Lawson, JD; Vanderpool, D; Wu, Y; Dougan, DR; Plonowski, A; Holub, C; Larson, C Discovery of potent, reversible MetAP2 inhibitors via fragment based drug discovery and structure based drug design-Part 1. Bioorg Med Chem Lett26:2774-2778 (2016) [PubMed] Article