| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 1A2 |
|---|
| Ligand | BDBM50169425 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1578011 (CHEMBL3806760) |
|---|
| IC50 | >9000±n/a nM |
|---|
| Citation |  Focken, T; Liu, S; Chahal, N; Dauphinais, M; Grimwood, ME; Chowdhury, S; Hemeon, I; Bichler, P; Bogucki, D; Waldbrook, M; Bankar, G; Sojo, LE; Young, C; Lin, S; Shuart, N; Kwan, R; Pang, J; Chang, JH; Safina, BS; Sutherlin, DP; Johnson, JP; Dehnhardt, CM; Mansour, TS; Oballa, RM; Cohen, CJ; Robinette, CL Discovery of Aryl Sulfonamides as Isoform-Selective Inhibitors of NaV1.7 with Efficacy in Rodent Pain Models. ACS Med Chem Lett7:277-82 (2016) [PubMed] Article Focken, T; Liu, S; Chahal, N; Dauphinais, M; Grimwood, ME; Chowdhury, S; Hemeon, I; Bichler, P; Bogucki, D; Waldbrook, M; Bankar, G; Sojo, LE; Young, C; Lin, S; Shuart, N; Kwan, R; Pang, J; Chang, JH; Safina, BS; Sutherlin, DP; Johnson, JP; Dehnhardt, CM; Mansour, TS; Oballa, RM; Cohen, CJ; Robinette, CL Discovery of Aryl Sulfonamides as Isoform-Selective Inhibitors of NaV1.7 with Efficacy in Rodent Pain Models. ACS Med Chem Lett7:277-82 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 1A2 |
|---|
| Name: | Cytochrome P450 1A2 |
|---|
| Synonyms: | CP1A2_HUMAN | CYP1A2 | CYPIA2 | Cholesterol 25-hydroxylase | Cytochrome P(3)450 | Cytochrome P450 1A | Cytochrome P450 1A2 (CYP1A2) | Cytochrome P450 4 | Cytochrome P450-P3 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 58423.38 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P05177 |
|---|
| Residue: | 516 |
|---|
| Sequence: | MALSQSVPFSATELLLASAIFCLVFWVLKGLRPRVPKGLKSPPEPWGWPLLGHVLTLGKN
PHLALSRMSQRYGDVLQIRIGSTPVLVLSRLDTIRQALVRQGDDFKGRPDLYTSTLITDG
QSLTFSTDSGPVWAARRRLAQNALNTFSIASDPASSSSCYLEEHVSKEAKALISRLQELM
AGPGHFDPYNQVVVSVANVIGAMCFGQHFPESSDEMLSLVKNTHEFVETASSGNPLDFFP
ILRYLPNPALQRFKAFNQRFLWFLQKTVQEHYQDFDKNSVRDITGALFKHSKKGPRASGN
LIPQEKIVNLVNDIFGAGFDTVTTAISWSLMYLVTKPEIQRKIQKELDTVIGRERRPRLS
DRPQLPYLEAFILETFRHSSFLPFTIPHSTTRDTTLNGFYIPKKCCVFVNQWQVNHDPEL
WEDPSEFRPERFLTADGTAINKPLSEKMMLFGMGKRRCIGEVLAKWEIFLFLAILLQQLE
FSVPPGVKVDLTPIYGLTMKHARCEHVQARLRFSIN
|
|
|
|---|
| BDBM50169425 |
|---|
| n/a |
|---|
| Name | BDBM50169425 |
|---|
| Synonyms: | CHEMBL3341983 | US9481677, 44 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H12ClF2N5O4S2 |
|---|
| Mol. Mass. | 535.931 |
|---|
| SMILES | Nc1noc2ccc(cc12)-c1cc(Cl)ccc1Oc1cc(F)c(cc1F)S(=O)(=O)Nc1ncns1 |
|---|
| Structure | 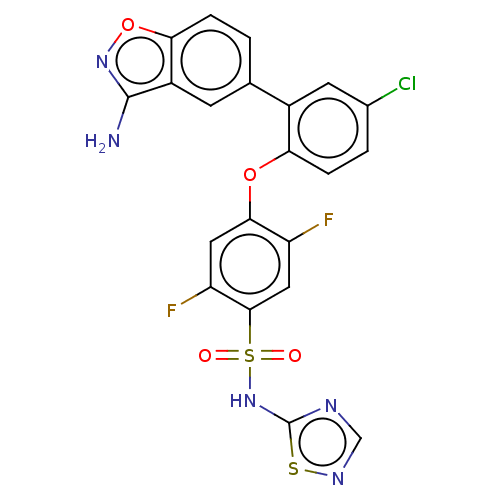
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Focken, T; Liu, S; Chahal, N; Dauphinais, M; Grimwood, ME; Chowdhury, S; Hemeon, I; Bichler, P; Bogucki, D; Waldbrook, M; Bankar, G; Sojo, LE; Young, C; Lin, S; Shuart, N; Kwan, R; Pang, J; Chang, JH; Safina, BS; Sutherlin, DP; Johnson, JP; Dehnhardt, CM; Mansour, TS; Oballa, RM; Cohen, CJ; Robinette, CL Discovery of Aryl Sulfonamides as Isoform-Selective Inhibitors of NaV1.7 with Efficacy in Rodent Pain Models. ACS Med Chem Lett7:277-82 (2016) [PubMed] Article
Focken, T; Liu, S; Chahal, N; Dauphinais, M; Grimwood, ME; Chowdhury, S; Hemeon, I; Bichler, P; Bogucki, D; Waldbrook, M; Bankar, G; Sojo, LE; Young, C; Lin, S; Shuart, N; Kwan, R; Pang, J; Chang, JH; Safina, BS; Sutherlin, DP; Johnson, JP; Dehnhardt, CM; Mansour, TS; Oballa, RM; Cohen, CJ; Robinette, CL Discovery of Aryl Sulfonamides as Isoform-Selective Inhibitors of NaV1.7 with Efficacy in Rodent Pain Models. ACS Med Chem Lett7:277-82 (2016) [PubMed] Article