| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Tyrosine-protein kinase ABL1 |
|---|
| Ligand | BDBM158154 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1577166 (CHEMBL3807428) |
|---|
| IC50 | >1000±n/a nM |
|---|
| Citation |  Menichincheri, M; Ardini, E; Magnaghi, P; Avanzi, N; Banfi, P; Bossi, R; Buffa, L; Canevari, G; Ceriani, L; Colombo, M; Corti, L; Donati, D; Fasolini, M; Felder, E; Fiorelli, C; Fiorentini, F; Galvani, A; Isacchi, A; Borgia, AL; Marchionni, C; Nesi, M; Orrenius, C; Panzeri, A; Pesenti, E; Rusconi, L; Saccardo, MB; Vanotti, E; Perrone, E; Orsini, P Discovery of Entrectinib: A New 3-Aminoindazole As a Potent Anaplastic Lymphoma Kinase (ALK), c-ros Oncogene 1 Kinase (ROS1), and Pan-Tropomyosin Receptor Kinases (Pan-TRKs) inhibitor. J Med Chem59:3392-408 (2016) [PubMed] Article Menichincheri, M; Ardini, E; Magnaghi, P; Avanzi, N; Banfi, P; Bossi, R; Buffa, L; Canevari, G; Ceriani, L; Colombo, M; Corti, L; Donati, D; Fasolini, M; Felder, E; Fiorelli, C; Fiorentini, F; Galvani, A; Isacchi, A; Borgia, AL; Marchionni, C; Nesi, M; Orrenius, C; Panzeri, A; Pesenti, E; Rusconi, L; Saccardo, MB; Vanotti, E; Perrone, E; Orsini, P Discovery of Entrectinib: A New 3-Aminoindazole As a Potent Anaplastic Lymphoma Kinase (ALK), c-ros Oncogene 1 Kinase (ROS1), and Pan-Tropomyosin Receptor Kinases (Pan-TRKs) inhibitor. J Med Chem59:3392-408 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Tyrosine-protein kinase ABL1 |
|---|
| Name: | Tyrosine-protein kinase ABL1 |
|---|
| Synonyms: | ABL | ABL1 | ABL1_HUMAN | Abelson murine leukemia viral oncogene homolog 1 | JTK7 | Proto-oncogene c-Abl | Proto-oncogene tyrosine-protein kinase ABL1 | Tyrosine-protein kinase (ABL) | Tyrosine-protein kinase ABL | Tyrosine-protein kinase ABL1 (ABL) | V-abl Abelson murine leukemia viral oncogene homolog 1 | c-ABL | p150 | tyrosine-protein kinase ABL1 isoform a |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 122897.30 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00519 |
|---|
| Residue: | 1130 |
|---|
| Sequence: | MLEICLKLVGCKSKKGLSSSSSCYLEEALQRPVASDFEPQGLSEAARWNSKENLLAGPSE
NDPNLFVALYDFVASGDNTLSITKGEKLRVLGYNHNGEWCEAQTKNGQGWVPSNYITPVN
SLEKHSWYHGPVSRNAAEYLLSSGINGSFLVRESESSPGQRSISLRYEGRVYHYRINTAS
DGKLYVSSESRFNTLAELVHHHSTVADGLITTLHYPAPKRNKPTVYGVSPNYDKWEMERT
DITMKHKLGGGQYGEVYEGVWKKYSLTVAVKTLKEDTMEVEEFLKEAAVMKEIKHPNLVQ
LLGVCTREPPFYIITEFMTYGNLLDYLRECNRQEVNAVVLLYMATQISSAMEYLEKKNFI
HRDLAARNCLVGENHLVKVADFGLSRLMTGDTYTAHAGAKFPIKWTAPESLAYNKFSIKS
DVWAFGVLLWEIATYGMSPYPGIDLSQVYELLEKDYRMERPEGCPEKVYELMRACWQWNP
SDRPSFAEIHQAFETMFQESSISDEVEKELGKQGVRGAVSTLLQAPELPTKTRTSRRAAE
HRDTTDVPEMPHSKGQGESDPLDHEPAVSPLLPRKERGPPEGGLNEDERLLPKDKKTNLF
SALIKKKKKTAPTPPKRSSSFREMDGQPERRGAGEEEGRDISNGALAFTPLDTADPAKSP
KPSNGAGVPNGALRESGGSGFRSPHLWKKSSTLTSSRLATGEEEGGGSSSKRFLRSCSAS
CVPHGAKDTEWRSVTLPRDLQSTGRQFDSSTFGGHKSEKPALPRKRAGENRSDQVTRGTV
TPPPRLVKKNEEAADEVFKDIMESSPGSSPPNLTPKPLRRQVTVAPASGLPHKEEAGKGS
ALGTPAAAEPVTPTSKAGSGAPGGTSKGPAEESRVRRHKHSSESPGRDKGKLSRLKPAPP
PPPAASAGKAGGKPSQSPSQEAAGEAVLGAKTKATSLVDAVNSDAAKPSQPGEGLKKPVL
PATPKPQSAKPSGTPISPAPVPSTLPSASSALAGDQPSSTAFIPLISTRVSLRKTRQPPE
RIASGAITKGVVLDSTEALCLAISRNSEQMASHSAVLEAGKNLYTFCVSYVDSIQQMRNK
FAFREAINKLENNLRELQICPATAGSGPAATQDFSKLLSSVKEISDIVQR
|
|
|
|---|
| BDBM158154 |
|---|
| n/a |
|---|
| Name | BDBM158154 |
|---|
| Synonyms: | US10081622, Compound 11 | US10370379, Entrectinib | US11059827, Compound Entrectinib | US11267806, Compound T-12 | US9029356, 11 | US9255087, 11 | US9616059, 11 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C31H34F2N6O2 |
|---|
| Mol. Mass. | 560.6375 |
|---|
| SMILES | CN1CCN(CC1)c1ccc(C(=O)Nc2n[nH]c3ccc(Cc4cc(F)cc(F)c4)cc23)c(NC2CCOCC2)c1 |
|---|
| Structure | 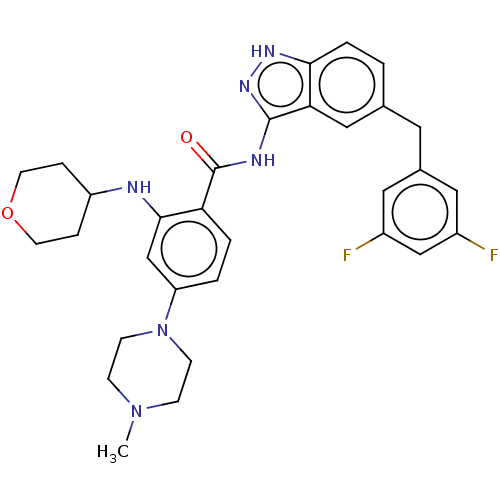
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Menichincheri, M; Ardini, E; Magnaghi, P; Avanzi, N; Banfi, P; Bossi, R; Buffa, L; Canevari, G; Ceriani, L; Colombo, M; Corti, L; Donati, D; Fasolini, M; Felder, E; Fiorelli, C; Fiorentini, F; Galvani, A; Isacchi, A; Borgia, AL; Marchionni, C; Nesi, M; Orrenius, C; Panzeri, A; Pesenti, E; Rusconi, L; Saccardo, MB; Vanotti, E; Perrone, E; Orsini, P Discovery of Entrectinib: A New 3-Aminoindazole As a Potent Anaplastic Lymphoma Kinase (ALK), c-ros Oncogene 1 Kinase (ROS1), and Pan-Tropomyosin Receptor Kinases (Pan-TRKs) inhibitor. J Med Chem59:3392-408 (2016) [PubMed] Article
Menichincheri, M; Ardini, E; Magnaghi, P; Avanzi, N; Banfi, P; Bossi, R; Buffa, L; Canevari, G; Ceriani, L; Colombo, M; Corti, L; Donati, D; Fasolini, M; Felder, E; Fiorelli, C; Fiorentini, F; Galvani, A; Isacchi, A; Borgia, AL; Marchionni, C; Nesi, M; Orrenius, C; Panzeri, A; Pesenti, E; Rusconi, L; Saccardo, MB; Vanotti, E; Perrone, E; Orsini, P Discovery of Entrectinib: A New 3-Aminoindazole As a Potent Anaplastic Lymphoma Kinase (ALK), c-ros Oncogene 1 Kinase (ROS1), and Pan-Tropomyosin Receptor Kinases (Pan-TRKs) inhibitor. J Med Chem59:3392-408 (2016) [PubMed] Article