| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cysteinyl leukotriene receptor 2 |
|---|
| Ligand | BDBM50009073 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1579541 (CHEMBL3810887) |
|---|
| IC50 | 58000±n/a nM |
|---|
| Citation |  Chen, H; Yang, H; Wang, Z; Xie, X; Nan, F Discovery of 3-Substituted 1H-Indole-2-carboxylic Acid Derivatives as a Novel Class of CysLT1 Selective Antagonists. ACS Med Chem Lett7:335-9 (2016) [PubMed] Article Chen, H; Yang, H; Wang, Z; Xie, X; Nan, F Discovery of 3-Substituted 1H-Indole-2-carboxylic Acid Derivatives as a Novel Class of CysLT1 Selective Antagonists. ACS Med Chem Lett7:335-9 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cysteinyl leukotriene receptor 2 |
|---|
| Name: | Cysteinyl leukotriene receptor 2 |
|---|
| Synonyms: | CLTR2_HUMAN | CYSLT2 | CYSLT2R | CYSLTR2 | Cysteinyl leukotriene receptor | Cysteinyl leukotriene receptor 2 | Leukotriene Cysteinyl 2 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 39657.52 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Leukotriene Cysteinyl 2 CYSLTR2 HUMAN::Q9NS75 |
|---|
| Residue: | 346 |
|---|
| Sequence: | MERKFMSLQPSISVSEMEPNGTFSNNNSRNCTIENFKREFFPIVYLIIFFWGVLGNGLSI
YVFLQPYKKSTSVNVFMLNLAISDLLFISTLPFRADYYLRGSNWIFGDLACRIMSYSLYV
NMYSSIYFLTVLSVVRFLAMVHPFRLLHVTSIRSAWILCGIIWILIMASSIMLLDSGSEQ
NGSVTSCLELNLYKIAKLQTMNYIALVVGCLLPFFTLSICYLLIIRVLLKVEVPESGLRV
SHRKALTTIIITLIIFFLCFLPYHTLRTVHLTTWKVGLCKDRLHKALVITLALAAANACF
NPLLYYFAGENFKDRLKSALRKGHPQKAKTKCVFPVSVWLRKETRV
|
|
|
|---|
| BDBM50009073 |
|---|
| n/a |
|---|
| Name | BDBM50009073 |
|---|
| Synonyms: | 4-(5-cyclopentyloxycarbonylamino-1-methyl-1H-indol-3-ylmethyl)-3-methoxy-N-o-tolylsulfonylbenzamide | CHEMBL603 | ZAFIRLUKAST | cyclopentyl 3-(2-methoxy-4-((o-tolylsulfonyl)carbamoyl)benzyl)-1-methylindole-5-carbamate | cyclopentyl 3-[2-methoxy-4-(2-methylphenylsulfonylcarbamoyl)benzyl]-1-methyl-1H-indol-5-ylcarbamate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C31H33N3O6S |
|---|
| Mol. Mass. | 575.675 |
|---|
| SMILES | COc1cc(ccc1Cc1cn(C)c2ccc(NC(=O)OC3CCCC3)cc12)C(=O)NS(=O)(=O)c1ccccc1C |
|---|
| Structure | 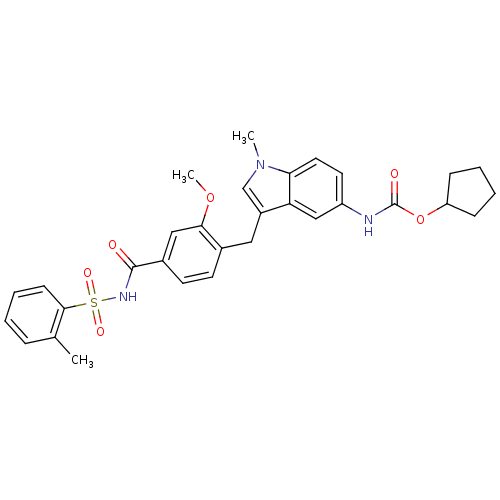
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Chen, H; Yang, H; Wang, Z; Xie, X; Nan, F Discovery of 3-Substituted 1H-Indole-2-carboxylic Acid Derivatives as a Novel Class of CysLT1 Selective Antagonists. ACS Med Chem Lett7:335-9 (2016) [PubMed] Article
Chen, H; Yang, H; Wang, Z; Xie, X; Nan, F Discovery of 3-Substituted 1H-Indole-2-carboxylic Acid Derivatives as a Novel Class of CysLT1 Selective Antagonists. ACS Med Chem Lett7:335-9 (2016) [PubMed] Article