| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Proto-oncogene tyrosine-protein kinase Src |
|---|
| Ligand | BDBM50189787 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1590291 (CHEMBL3830183) |
|---|
| IC50 | >100±n/a nM |
|---|
| Citation |  Yan, W; Wang, X; Dai, Y; Zhao, B; Yang, X; Fan, J; Gao, Y; Meng, F; Wang, Y; Luo, C; Ai, J; Geng, M; Duan, W Discovery of 3-(5'-Substituted)-Benzimidazole-5-(1-(3,5-dichloropyridin-4-yl)ethoxy)-1H-indazoles as Potent Fibroblast Growth Factor Receptor Inhibitors: Design, Synthesis, and Biological Evaluation. J Med Chem59:6690-708 (2016) [PubMed] Article Yan, W; Wang, X; Dai, Y; Zhao, B; Yang, X; Fan, J; Gao, Y; Meng, F; Wang, Y; Luo, C; Ai, J; Geng, M; Duan, W Discovery of 3-(5'-Substituted)-Benzimidazole-5-(1-(3,5-dichloropyridin-4-yl)ethoxy)-1H-indazoles as Potent Fibroblast Growth Factor Receptor Inhibitors: Design, Synthesis, and Biological Evaluation. J Med Chem59:6690-708 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Proto-oncogene tyrosine-protein kinase Src |
|---|
| Name: | Proto-oncogene tyrosine-protein kinase Src |
|---|
| Synonyms: | Calmodulin/Proto-oncogene tyrosine-protein kinase Src | Protein cereblon/Tyrosine-protein kinase SRC | Proto-oncogene c-Src | Proto-oncogene tyrosine-protein kinase Src (c-Src) | SRC | SRC1 | SRC_HUMAN | Tyrosine-protein kinase Src (SRC) | V-src sarcoma (Schmidt-Ruppin A-2) viral oncogene homolog (avian) | c-Src | p60-Src | pp60c-src |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 59838.60 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P12931 |
|---|
| Residue: | 536 |
|---|
| Sequence: | MGSNKSKPKDASQRRRSLEPAENVHGAGGGAFPASQTPSKPASADGHRGPSAAFAPAAAE
PKLFGGFNSSDTVTSPQRAGPLAGGVTTFVALYDYESRTETDLSFKKGERLQIVNNTEGD
WWLAHSLSTGQTGYIPSNYVAPSDSIQAEEWYFGKITRRESERLLLNAENPRGTFLVRES
ETTKGAYCLSVSDFDNAKGLNVKHYKIRKLDSGGFYITSRTQFNSLQQLVAYYSKHADGL
CHRLTTVCPTSKPQTQGLAKDAWEIPRESLRLEVKLGQGCFGEVWMGTWNGTTRVAIKTL
KPGTMSPEAFLQEAQVMKKLRHEKLVQLYAVVSEEPIYIVTEYMSKGSLLDFLKGETGKY
LRLPQLVDMAAQIASGMAYVERMNYVHRDLRAANILVGENLVCKVADFGLARLIEDNEYT
ARQGAKFPIKWTAPEAALYGRFTIKSDVWSFGILLTELTTKGRVPYPGMVNREVLDQVER
GYRMPCPPECPESLHDLMCQCWRKEPEERPTFEYLQAFLEDYFTSTEPQYQPGENL
|
|
|
|---|
| BDBM50189787 |
|---|
| n/a |
|---|
| Name | BDBM50189787 |
|---|
| Synonyms: | CHEMBL3827178 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H25Cl2N7O |
|---|
| Mol. Mass. | 522.429 |
|---|
| SMILES | C[C@@H](Oc1ccc2[nH]nc(-c3nc4cc(ccc4[nH]3)N3CCN(C)CC3)c2c1)c1c(Cl)cncc1Cl |r| |
|---|
| Structure | 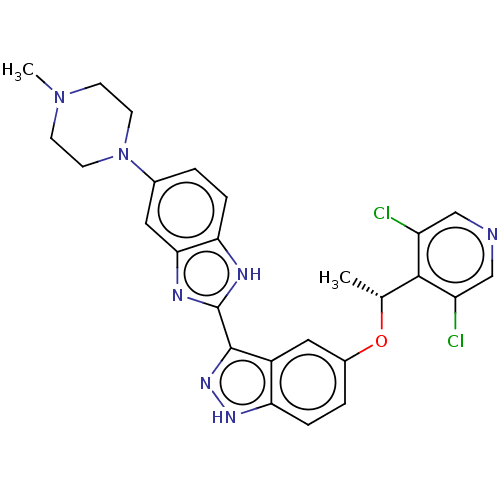
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Yan, W; Wang, X; Dai, Y; Zhao, B; Yang, X; Fan, J; Gao, Y; Meng, F; Wang, Y; Luo, C; Ai, J; Geng, M; Duan, W Discovery of 3-(5'-Substituted)-Benzimidazole-5-(1-(3,5-dichloropyridin-4-yl)ethoxy)-1H-indazoles as Potent Fibroblast Growth Factor Receptor Inhibitors: Design, Synthesis, and Biological Evaluation. J Med Chem59:6690-708 (2016) [PubMed] Article
Yan, W; Wang, X; Dai, Y; Zhao, B; Yang, X; Fan, J; Gao, Y; Meng, F; Wang, Y; Luo, C; Ai, J; Geng, M; Duan, W Discovery of 3-(5'-Substituted)-Benzimidazole-5-(1-(3,5-dichloropyridin-4-yl)ethoxy)-1H-indazoles as Potent Fibroblast Growth Factor Receptor Inhibitors: Design, Synthesis, and Biological Evaluation. J Med Chem59:6690-708 (2016) [PubMed] Article