| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cathepsin D |
|---|
| Ligand | BDBM50195235 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1617588 (CHEMBL3859657) |
|---|
| IC50 | >20000±n/a nM |
|---|
| Citation |  Connolly, S; L�nn, H; K�ck, H; Van de Po�l, A; Dahl, G; Doyle, K; Gardiner, P; Root, J; Wikell, C; Johannesson, P; Stenvall, K; Swallow, S Discovery of Second Generation Reversible Covalent DPP1 Inhibitors Leading to an Oxazepane Amidoacetonitrile Based Clinical Candidate (AZD7986). J Med Chem59:9457-9472 (2016) [PubMed] Article Connolly, S; L�nn, H; K�ck, H; Van de Po�l, A; Dahl, G; Doyle, K; Gardiner, P; Root, J; Wikell, C; Johannesson, P; Stenvall, K; Swallow, S Discovery of Second Generation Reversible Covalent DPP1 Inhibitors Leading to an Oxazepane Amidoacetonitrile Based Clinical Candidate (AZD7986). J Med Chem59:9457-9472 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cathepsin D |
|---|
| Name: | Cathepsin D |
|---|
| Synonyms: | CATD_HUMAN | CPSD | CTSD | Cathepsin D [Precursor] | Cathepsin D heavy chain | Cathepsin D light chain | Cathepsin D precursor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 44551.72 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Human proCathepsin D (SwissProt accession number P07339) was expressed in Sf9 cells, purified, and autoactivated. |
|---|
| Residue: | 412 |
|---|
| Sequence: | MQPSSLLPLALCLLAAPASALVRIPLHKFTSIRRTMSEVGGSVEDLIAKGPVSKYSQAVP
AVTEGPIPEVLKNYMDAQYYGEIGIGTPPQCFTVVFDTGSSNLWVPSIHCKLLDIACWIH
HKYNSDKSSTYVKNGTSFDIHYGSGSLSGYLSQDTVSVPCQSASSASALGGVKVERQVFG
EATKQPGITFIAAKFDGILGMAYPRISVNNVLPVFDNLMQQKLVDQNIFSFYLSRDPDAQ
PGGELMLGGTDSKYYKGSLSYLNVTRKAYWQVHLDQVEVASGLTLCKEGCEAIVDTGTSL
MVGPVDEVRELQKAIGAVPLIQGEYMIPCEKVSTLPAITLKLGGKGYKLSPEDYTLKVSQ
AGKTLCLSGFMGMDIPPPSGPLWILGDVFIGRYYTVFDRDNNRVGFAEAARL
|
|
|
|---|
| BDBM50195235 |
|---|
| n/a |
|---|
| Name | BDBM50195235 |
|---|
| Synonyms: | CHEMBL3900409 | US10287258, Example 2 | US10669245, Example 2 | US11117874, Example 2 | US11655221, Example 2 | US11680049, Example 2 | US20230278969, Example 2 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H24N4O4 |
|---|
| Mol. Mass. | 420.4611 |
|---|
| SMILES | Cn1c2cc(ccc2oc1=O)-c1ccc(C[C@H](NC(=O)[C@@H]2CNCCCO2)C#N)cc1 |r| |
|---|
| Structure | 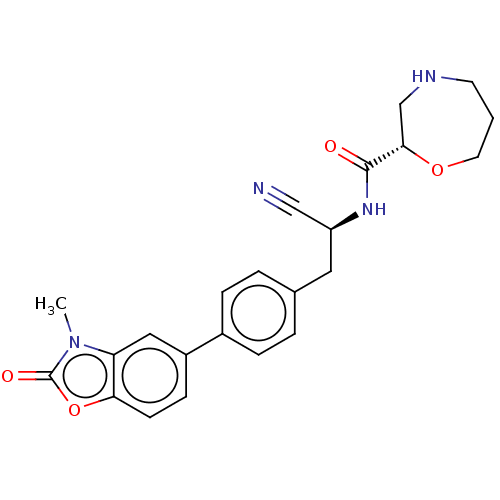
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Connolly, S; L�nn, H; K�ck, H; Van de Po�l, A; Dahl, G; Doyle, K; Gardiner, P; Root, J; Wikell, C; Johannesson, P; Stenvall, K; Swallow, S Discovery of Second Generation Reversible Covalent DPP1 Inhibitors Leading to an Oxazepane Amidoacetonitrile Based Clinical Candidate (AZD7986). J Med Chem59:9457-9472 (2016) [PubMed] Article
Connolly, S; L�nn, H; K�ck, H; Van de Po�l, A; Dahl, G; Doyle, K; Gardiner, P; Root, J; Wikell, C; Johannesson, P; Stenvall, K; Swallow, S Discovery of Second Generation Reversible Covalent DPP1 Inhibitors Leading to an Oxazepane Amidoacetonitrile Based Clinical Candidate (AZD7986). J Med Chem59:9457-9472 (2016) [PubMed] Article