| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Alpha-1D adrenergic receptor |
|---|
| Ligand | BDBM50160154 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1617622 (CHEMBL3859691) |
|---|
| IC50 | 6.3±n/a nM |
|---|
| Citation |  Guo, D; Li, J; Lin, H; Zhou, Y; Chen, Y; Zhao, F; Sun, H; Zhang, D; Li, H; Shoichet, BK; Shan, L; Zhang, W; Xie, X; Jiang, H; Liu, H Design, Synthesis, and Biological Evaluation of Novel Tetrahydroprotoberberine Derivatives (THPBs) as Selectivea J Med Chem59:9489-9502 (2016) [PubMed] Article Guo, D; Li, J; Lin, H; Zhou, Y; Chen, Y; Zhao, F; Sun, H; Zhang, D; Li, H; Shoichet, BK; Shan, L; Zhang, W; Xie, X; Jiang, H; Liu, H Design, Synthesis, and Biological Evaluation of Novel Tetrahydroprotoberberine Derivatives (THPBs) as Selectivea J Med Chem59:9489-9502 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Alpha-1D adrenergic receptor |
|---|
| Name: | Alpha-1D adrenergic receptor |
|---|
| Synonyms: | ADA1D_HUMAN | ADRA1A | ADRA1D | Adrenergic receptor | Adrenergic receptor alpha | Alpha 1D-adrenoceptor | Alpha 1D-adrenoreceptor | Alpha adrenergic receptor (1a and 1d) | Alpha-1D adrenoceptor | Alpha-adrenergic receptor 1a | adrenergic Alpha1D |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 60485.82 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | adrenergic Alpha1D ADRA1D HUMAN::P25100 |
|---|
| Residue: | 572 |
|---|
| Sequence: | MTFRDLLSVSFEGPRPDSSAGGSSAGGGGGSAGGAAPSEGPAVGGVPGGAGGGGGVVGAG
SGEDNRSSAGEPGSAGAGGDVNGTAAVGGLVVSAQGVGVGVFLAAFILMAVAGNLLVILS
VACNRHLQTVTNYFIVNLAVADLLLSATVLPFSATMEVLGFWAFGRAFCDVWAAVDVLCC
TASILSLCTISVDRYVGVRHSLKYPAIMTERKAAAILALLWVVALVVSVGPLLGWKEPVP
PDERFCGITEEAGYAVFSSVCSFYLPMAVIVVMYCRVYVVARSTTRSLEAGVKRERGKAS
EVVLRIHCRGAATGADGAHGMRSAKGHTFRSSLSVRLLKFSREKKAAKTLAIVVGVFVLC
WFPFFFVLPLGSLFPQLKPSEGVFKVIFWLGYFNSCVNPLIYPCSSREFKRAFLRLLRCQ
CRRRRRRRPLWRVYGHHWRASTSGLRQDCAPSSGDAPPGAPLALTALPDPDPEPPGTPEM
QAPVASRRKPPSAFREWRLLGPFRRPTTQLRAKVSSLSHKIRAGGAQRAEAACAQRSEVE
AVSLGVPHEVAEGATCQAYELADYSNLRETDI
|
|
|
|---|
| BDBM50160154 |
|---|
| n/a |
|---|
| Name | BDBM50160154 |
|---|
| Synonyms: | 1-(3-Hydroxy-propyl)-5-((R)-2-{2-[2-(2,2,2-trifluoro-ethoxy)-phenoxy]-ethylamino}-propyl)-2,3-dihydro-1H-indole-7-carboxylic acid amide | CHEMBL24778 | SILODOSIN |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H32F3N3O4 |
|---|
| Mol. Mass. | 495.5345 |
|---|
| SMILES | C[C@H](Cc1cc2CCN(CCCO)c2c(c1)C(N)=O)NCCOc1ccccc1OCC(F)(F)F |
|---|
| Structure | 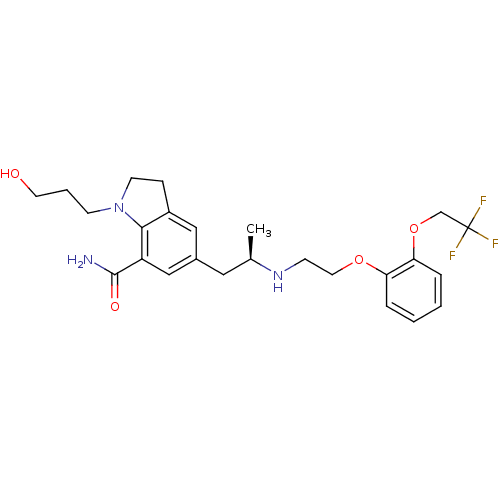
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Guo, D; Li, J; Lin, H; Zhou, Y; Chen, Y; Zhao, F; Sun, H; Zhang, D; Li, H; Shoichet, BK; Shan, L; Zhang, W; Xie, X; Jiang, H; Liu, H Design, Synthesis, and Biological Evaluation of Novel Tetrahydroprotoberberine Derivatives (THPBs) as Selectivea J Med Chem59:9489-9502 (2016) [PubMed] Article
Guo, D; Li, J; Lin, H; Zhou, Y; Chen, Y; Zhao, F; Sun, H; Zhang, D; Li, H; Shoichet, BK; Shan, L; Zhang, W; Xie, X; Jiang, H; Liu, H Design, Synthesis, and Biological Evaluation of Novel Tetrahydroprotoberberine Derivatives (THPBs) as Selectivea J Med Chem59:9489-9502 (2016) [PubMed] Article