| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Epidermal growth factor receptor |
|---|
| Ligand | BDBM149404 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1618202 (CHEMBL3860371) |
|---|
| IC50 | 262±n/a nM |
|---|
| Citation |  Lelais, G; Epple, R; Marsilje, TH; Long, YO; McNeill, M; Chen, B; Lu, W; Anumolu, J; Badiger, S; Bursulaya, B; DiDonato, M; Fong, R; Juarez, J; Li, J; Manuia, M; Mason, DE; Gordon, P; Groessl, T; Johnson, K; Jia, Y; Kasibhatla, S; Li, C; Isbell, J; Spraggon, G; Bender, S; Michellys, PY Discovery of (R,E)-N-(7-Chloro-1-(1-[4-(dimethylamino)but-2-enoyl]azepan-3-yl)-1H-benzo[d]imidazol-2-yl)-2-methylisonicotinamide (EGF816), a Novel, Potent, and WT Sparing Covalent Inhibitor of Oncogenic (L858R, ex19del) and Resistant (T790M) EGFR Mutants for the Treatment of EGFR Mutant Non-Small-Ce J Med Chem59:6671-89 (2016) [PubMed] Article Lelais, G; Epple, R; Marsilje, TH; Long, YO; McNeill, M; Chen, B; Lu, W; Anumolu, J; Badiger, S; Bursulaya, B; DiDonato, M; Fong, R; Juarez, J; Li, J; Manuia, M; Mason, DE; Gordon, P; Groessl, T; Johnson, K; Jia, Y; Kasibhatla, S; Li, C; Isbell, J; Spraggon, G; Bender, S; Michellys, PY Discovery of (R,E)-N-(7-Chloro-1-(1-[4-(dimethylamino)but-2-enoyl]azepan-3-yl)-1H-benzo[d]imidazol-2-yl)-2-methylisonicotinamide (EGF816), a Novel, Potent, and WT Sparing Covalent Inhibitor of Oncogenic (L858R, ex19del) and Resistant (T790M) EGFR Mutants for the Treatment of EGFR Mutant Non-Small-Ce J Med Chem59:6671-89 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Epidermal growth factor receptor |
|---|
| Name: | Epidermal growth factor receptor |
|---|
| Synonyms: | EGF Receptor Substrate | EGFR | EGFR_HUMAN | ERBB | ERBB1 | Epidermal Growth Factor Receptor Tyrosine Kinase | Epidermal Growth Factor Receptor erbB-1 | Epidermal Growth Factor Receptor, ErbB-1 | Epidermal growth factor receptor (EGFR) | Epidermal growth factor receptor (HER1) | HER1 | Protein-Tyrosine Kinase Erbb-1 | Proto-oncogene c-ErbB-1 | Receptor tyrosine-protein kinase ErbB-1 | Tyrosine-Protein Kinase ErbB-1 |
|---|
| Type: | Receptor Kinase Domain |
|---|
| Mol. Mass.: | 134279.59 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00533 |
|---|
| Residue: | 1210 |
|---|
| Sequence: | MRPSGTAGAALLALLAALCPASRALEEKKVCQGTSNKLTQLGTFEDHFLSLQRMFNNCEV
VLGNLEITYVQRNYDLSFLKTIQEVAGYVLIALNTVERIPLENLQIIRGNMYYENSYALA
VLSNYDANKTGLKELPMRNLQEILHGAVRFSNNPALCNVESIQWRDIVSSDFLSNMSMDF
QNHLGSCQKCDPSCPNGSCWGAGEENCQKLTKIICAQQCSGRCRGKSPSDCCHNQCAAGC
TGPRESDCLVCRKFRDEATCKDTCPPLMLYNPTTYQMDVNPEGKYSFGATCVKKCPRNYV
VTDHGSCVRACGADSYEMEEDGVRKCKKCEGPCRKVCNGIGIGEFKDSLSINATNIKHFK
NCTSISGDLHILPVAFRGDSFTHTPPLDPQELDILKTVKEITGFLLIQAWPENRTDLHAF
ENLEIIRGRTKQHGQFSLAVVSLNITSLGLRSLKEISDGDVIISGNKNLCYANTINWKKL
FGTSGQKTKIISNRGENSCKATGQVCHALCSPEGCWGPEPRDCVSCRNVSRGRECVDKCN
LLEGEPREFVENSECIQCHPECLPQAMNITCTGRGPDNCIQCAHYIDGPHCVKTCPAGVM
GENNTLVWKYADAGHVCHLCHPNCTYGCTGPGLEGCPTNGPKIPSIATGMVGALLLLLVV
ALGIGLFMRRRHIVRKRTLRRLLQERELVEPLTPSGEAPNQALLRILKETEFKKIKVLGS
GAFGTVYKGLWIPEGEKVKIPVAIKELREATSPKANKEILDEAYVMASVDNPHVCRLLGI
CLTSTVQLITQLMPFGCLLDYVREHKDNIGSQYLLNWCVQIAKGMNYLEDRRLVHRDLAA
RNVLVKTPQHVKITDFGLAKLLGAEEKEYHAEGGKVPIKWMALESILHRIYTHQSDVWSY
GVTVWELMTFGSKPYDGIPASEISSILEKGERLPQPPICTIDVYMIMVKCWMIDADSRPK
FRELIIEFSKMARDPQRYLVIQGDERMHLPSPTDSNFYRALMDEEDMDDVVDADEYLIPQ
QGFFSSPSTSRTPLLSSLSATSNNSTVACIDRNGLQSCPIKEDSFLQRYSSDPTGALTED
SIDDTFLPVPEYINQSVPKRPAGSVQNPVYHNQPLNPAPSRDPHYQDPHSTAVGNPEYLN
TVQPTCVNSTFDSPAHWAQKGSHQISLDNPDYQQDFFPKEAKPNGIFKGSTAENAEYLRV
APQSSEFIGA
|
|
|
|---|
| BDBM149404 |
|---|
| n/a |
|---|
| Name | BDBM149404 |
|---|
| Synonyms: | AVL-301 | CHEMBL3545308 | CNX-419 | CO-1686 | Rociletinib | Tube721 | US10167264, Compound CO-1686 | US10300058, CO-1686 | US10434101, Compound I-4 | US11096942, Compound I-4 | US8975249, I-4 | US9783524, CO-1686 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H28F3N7O3 |
|---|
| Mol. Mass. | 555.5515 |
|---|
| SMILES | COc1cc(ccc1Nc1ncc(c(Nc2cccc(NC(=O)C=C)c2)n1)C(F)(F)F)N1CCN(CC1)C(C)=O |
|---|
| Structure | 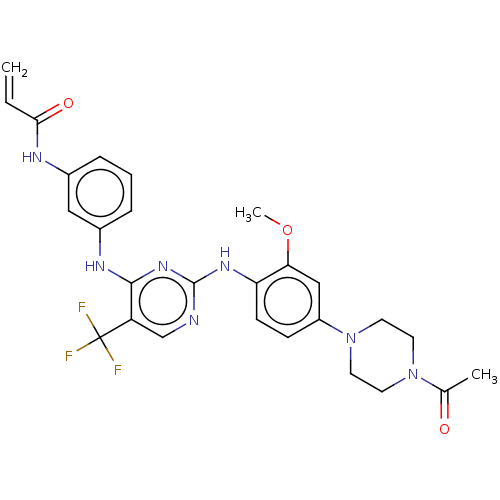
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Lelais, G; Epple, R; Marsilje, TH; Long, YO; McNeill, M; Chen, B; Lu, W; Anumolu, J; Badiger, S; Bursulaya, B; DiDonato, M; Fong, R; Juarez, J; Li, J; Manuia, M; Mason, DE; Gordon, P; Groessl, T; Johnson, K; Jia, Y; Kasibhatla, S; Li, C; Isbell, J; Spraggon, G; Bender, S; Michellys, PY Discovery of (R,E)-N-(7-Chloro-1-(1-[4-(dimethylamino)but-2-enoyl]azepan-3-yl)-1H-benzo[d]imidazol-2-yl)-2-methylisonicotinamide (EGF816), a Novel, Potent, and WT Sparing Covalent Inhibitor of Oncogenic (L858R, ex19del) and Resistant (T790M) EGFR Mutants for the Treatment of EGFR Mutant Non-Small-Ce J Med Chem59:6671-89 (2016) [PubMed] Article
Lelais, G; Epple, R; Marsilje, TH; Long, YO; McNeill, M; Chen, B; Lu, W; Anumolu, J; Badiger, S; Bursulaya, B; DiDonato, M; Fong, R; Juarez, J; Li, J; Manuia, M; Mason, DE; Gordon, P; Groessl, T; Johnson, K; Jia, Y; Kasibhatla, S; Li, C; Isbell, J; Spraggon, G; Bender, S; Michellys, PY Discovery of (R,E)-N-(7-Chloro-1-(1-[4-(dimethylamino)but-2-enoyl]azepan-3-yl)-1H-benzo[d]imidazol-2-yl)-2-methylisonicotinamide (EGF816), a Novel, Potent, and WT Sparing Covalent Inhibitor of Oncogenic (L858R, ex19del) and Resistant (T790M) EGFR Mutants for the Treatment of EGFR Mutant Non-Small-Ce J Med Chem59:6671-89 (2016) [PubMed] Article