| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Histone-lysine N-methyltransferase EZH1 |
|---|
| Ligand | BDBM50205639 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1628507 (CHEMBL3871092) |
|---|
| IC50 | 3340±n/a nM |
|---|
| Citation |  Ansari, A; Satalkar, S; Patil, V; Shete, AS; Kaur, S; Gupta, A; Singh, S; Raja, M; Severance, DL; Bernales, S; Chakravarty, S; Hung, DT; Pham, SM; Herrera, FJ; Rai, R Novel 3-methylindoline inhibitors of EZH2: Design, synthesis and SAR. Bioorg Med Chem Lett27:217-222 (2017) [PubMed] Article Ansari, A; Satalkar, S; Patil, V; Shete, AS; Kaur, S; Gupta, A; Singh, S; Raja, M; Severance, DL; Bernales, S; Chakravarty, S; Hung, DT; Pham, SM; Herrera, FJ; Rai, R Novel 3-methylindoline inhibitors of EZH2: Design, synthesis and SAR. Bioorg Med Chem Lett27:217-222 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Histone-lysine N-methyltransferase EZH1 |
|---|
| Name: | Histone-lysine N-methyltransferase EZH1 |
|---|
| Synonyms: | ENX-2 | EZH1 | EZH1_HUMAN | Enhancer of zeste homolog 1 | Enhancer of zeste homolog 1 (EZH1) | Histone-lysine N-methyltransferase EZH1 | KIAA0388 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 85285.34 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q92800 |
|---|
| Residue: | 747 |
|---|
| Sequence: | MEIPNPPTSKCITYWKRKVKSEYMRLRQLKRLQANMGAKALYVANFAKVQEKTQILNEEW
KKLRVQPVQSMKPVSGHPFLKKCTIESIFPGFASQHMLMRSLNTVALVPIMYSWSPLQQN
FMVEDETVLCNIPYMGDEVKEEDETFIEELINNYDGKVHGEEEMIPGSVLISDAVFLELV
DALNQYSDEEEEGHNDTSDGKQDDSKEDLPVTRKRKRHAIEGNKKSSKKQFPNDMIFSAI
ASMFPENGVPDDMKERYRELTEMSDPNALPPQCTPNIDGPNAKSVQREQSLHSFHTLFCR
RCFKYDCFLHPFHATPNVYKRKNKEIKIEPEPCGTDCFLLLEGAKEYAMLHNPRSKCSGR
RRRRHHIVSASCSNASASAVAETKEGDSDRDTGNDWASSSSEANSRCQTPTKQKASPAPP
QLCVVEAPSEPVEWTGAEESLFRVFHGTYFNNFCSIARLLGTKTCKQVFQFAVKESLILK
LPTDELMNPSQKKKRKHRLWAAHCRKIQLKKDNSSTQVYNYQPCDHPDRPCDSTCPCIMT
QNFCEKFCQCNPDCQNRFPGCRCKTQCNTKQCPCYLAVRECDPDLCLTCGASEHWDCKVV
SCKNCSIQRGLKKHLLLAPSDVAGWGTFIKESVQKNEFISEYCGELISQDEADRRGKVYD
KYMSSFLFNLNNDFVVDATRKGNKIRFANHSVNPNCYAKVVMVNGDHRIGIFAKRAIQAG
EELFFDYRYSQADALKYVGIERETDVL
|
|
|
|---|
| BDBM50205639 |
|---|
| n/a |
|---|
| Name | BDBM50205639 |
|---|
| Synonyms: | CHEMBL3892623 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C34H42N4O4 |
|---|
| Mol. Mass. | 570.7217 |
|---|
| SMILES | CC1CN(C2CCOCC2)c2cc(cc(C(=O)NCc3c(C)cc(C)[nH]c3=O)c12)-c1ccc(CN2CCOCC2)cc1 |
|---|
| Structure | 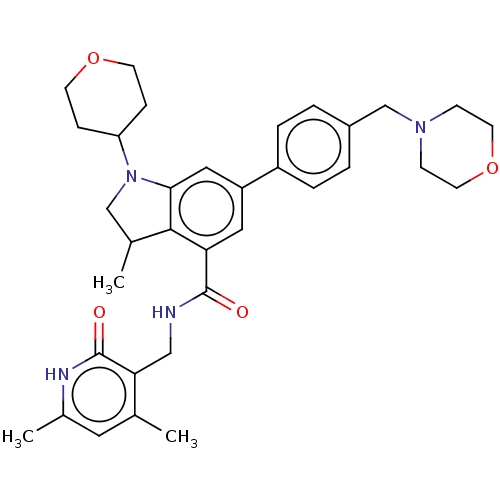
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ansari, A; Satalkar, S; Patil, V; Shete, AS; Kaur, S; Gupta, A; Singh, S; Raja, M; Severance, DL; Bernales, S; Chakravarty, S; Hung, DT; Pham, SM; Herrera, FJ; Rai, R Novel 3-methylindoline inhibitors of EZH2: Design, synthesis and SAR. Bioorg Med Chem Lett27:217-222 (2017) [PubMed] Article
Ansari, A; Satalkar, S; Patil, V; Shete, AS; Kaur, S; Gupta, A; Singh, S; Raja, M; Severance, DL; Bernales, S; Chakravarty, S; Hung, DT; Pham, SM; Herrera, FJ; Rai, R Novel 3-methylindoline inhibitors of EZH2: Design, synthesis and SAR. Bioorg Med Chem Lett27:217-222 (2017) [PubMed] Article