| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Neuromedin-K receptor |
|---|
| Ligand | BDBM50051295 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1633582 (CHEMBL3876374) |
|---|
| IC50 | 6.7±n/a nM |
|---|
| Citation |  Gunia-Krzyzak, A; Zelaszczyk, D; Rapacz, A; Zeslawska, E; Waszkielewicz, AM; Panczyk, K; Sloczynska, K; Pekala, E; Nitek, W; Filipek, B; Marona, H Structure-anticonvulsant activity studies in the group of (E)-N-cinnamoyl aminoalkanols derivatives monosubstituted in phenyl ring with 4-Cl, 4-CH Bioorg Med Chem25:471-482 (2017) [PubMed] Article Gunia-Krzyzak, A; Zelaszczyk, D; Rapacz, A; Zeslawska, E; Waszkielewicz, AM; Panczyk, K; Sloczynska, K; Pekala, E; Nitek, W; Filipek, B; Marona, H Structure-anticonvulsant activity studies in the group of (E)-N-cinnamoyl aminoalkanols derivatives monosubstituted in phenyl ring with 4-Cl, 4-CH Bioorg Med Chem25:471-482 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Neuromedin-K receptor |
|---|
| Name: | Neuromedin-K receptor |
|---|
| Synonyms: | NK-3 receptor | NK-3R | NK3R | NK3R_HUMAN | NKR | Neurokinin 3 receptor | Neurokinin B receptor | Neurokinin-3 (NK-3) | Neuromedin-3 receptor (NK-3R) | Neuromedin-3 receptor (NK3) | Neuromedin-K receptor | Neuromedin-K receptor (NK-3 receptor) | Neuromedin-K receptor (NK3) | Neuromedin-K receptor(NK3R) | TAC3R | TACR3 | Tachykinin receptor 3 | Tachykinin receptor 3 (NK3) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 52221.96 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P29371 |
|---|
| Residue: | 465 |
|---|
| Sequence: | MATLPAAETWIDGGGGVGADAVNLTASLAAGAATGAVETGWLQLLDQAGNLSSSPSALGL
PVASPAPSQPWANLTNQFVQPSWRIALWSLAYGVVVAVAVLGNLIVIWIILAHKRMRTVT
NYFLVNLAFSDASMAAFNTLVNFIYALHSEWYFGANYCRFQNFFPITAVFASIYSMTAIA
VDRYMAIIDPLKPRLSATATKIVIGSIWILAFLLAFPQCLYSKTKVMPGRTLCFVQWPEG
PKQHFTYHIIVIILVYCFPLLIMGITYTIVGITLWGGEIPGDTCDKYHEQLKAKRKVVKM
MIIVVMTFAICWLPYHIYFILTAIYQQLNRWKYIQQVYLASFWLAMSSTMYNPIIYCCLN
KRFRAGFKRAFRWCPFIKVSSYDELELKTTRFHPNRQSSMYTVTRMESMTVVFDPNDADT
TRSSRKKRATPRDPSFNGCSRRNSKSASATSSFISSPYTSVDEYS
|
|
|
|---|
| BDBM50051295 |
|---|
| n/a |
|---|
| Name | BDBM50051295 |
|---|
| Synonyms: | (S)-3-methyl-2-phenyl-N-(1-phenylpropyl)quinoline-4-carboxamide | 3-Methyl-2-phenyl-quinoline-4-carboxylic acid ((S)-1-phenyl-propyl)-amide | CHEMBL10284 | SB-222200 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H24N2O |
|---|
| Mol. Mass. | 380.4816 |
|---|
| SMILES | CC[C@H](NC(=O)c1c(C)c(nc2ccccc12)-c1ccccc1)c1ccccc1 |r| |
|---|
| Structure | 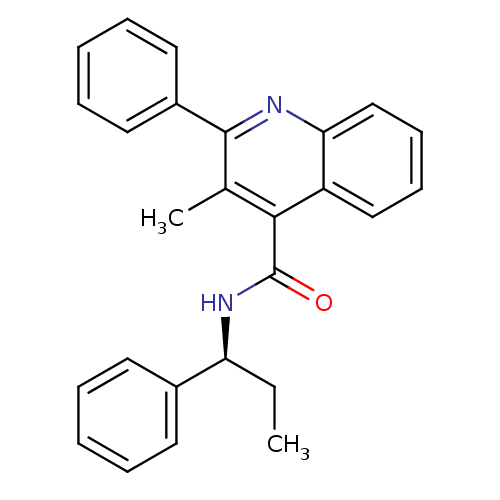
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Gunia-Krzyzak, A; Zelaszczyk, D; Rapacz, A; Zeslawska, E; Waszkielewicz, AM; Panczyk, K; Sloczynska, K; Pekala, E; Nitek, W; Filipek, B; Marona, H Structure-anticonvulsant activity studies in the group of (E)-N-cinnamoyl aminoalkanols derivatives monosubstituted in phenyl ring with 4-Cl, 4-CH Bioorg Med Chem25:471-482 (2017) [PubMed] Article
Gunia-Krzyzak, A; Zelaszczyk, D; Rapacz, A; Zeslawska, E; Waszkielewicz, AM; Panczyk, K; Sloczynska, K; Pekala, E; Nitek, W; Filipek, B; Marona, H Structure-anticonvulsant activity studies in the group of (E)-N-cinnamoyl aminoalkanols derivatives monosubstituted in phenyl ring with 4-Cl, 4-CH Bioorg Med Chem25:471-482 (2017) [PubMed] Article