| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Tyrosine-protein kinase Lck |
|---|
| Ligand | BDBM50211223 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1636092 (CHEMBL3878990) |
|---|
| IC50 | 12±n/a nM |
|---|
| Citation |  Wang, Q; Liu, F; Wang, B; Zou, F; Qi, Z; Chen, C; Yu, K; Hu, C; Qi, S; Wang, W; Hu, Z; Liu, J; Wang, W; Wang, L; Liang, Q; Zhang, S; Ren, T; Liu, Q; Liu, J Discovery of 4-Methyl-N-(4-((4-methylpiperazin-1-yl)methyl)-3-(trifluoromethyl)phenyl)-3-((1-nicotinoylpiperidin-4-yl)oxy)benzamide (CHMFL-ABL/KIT-155) as a Novel Highly Potent Type II ABL/KIT Dual Kinase Inhibitor with a Distinct Hinge Binding. J Med Chem60:273-289 (2017) [PubMed] Article Wang, Q; Liu, F; Wang, B; Zou, F; Qi, Z; Chen, C; Yu, K; Hu, C; Qi, S; Wang, W; Hu, Z; Liu, J; Wang, W; Wang, L; Liang, Q; Zhang, S; Ren, T; Liu, Q; Liu, J Discovery of 4-Methyl-N-(4-((4-methylpiperazin-1-yl)methyl)-3-(trifluoromethyl)phenyl)-3-((1-nicotinoylpiperidin-4-yl)oxy)benzamide (CHMFL-ABL/KIT-155) as a Novel Highly Potent Type II ABL/KIT Dual Kinase Inhibitor with a Distinct Hinge Binding. J Med Chem60:273-289 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Tyrosine-protein kinase Lck |
|---|
| Name: | Tyrosine-protein kinase Lck |
|---|
| Synonyms: | 2.7.10.2 | LCK | LCK_HUMAN | LSK | Leukocyte C-terminal Src kinase | Lymphocyte cell-specific protein-tyrosine kinase | Lymphocyte-specific protein tyrosine kinase | P56-LCK | Protein YT16 | Proto-oncogene Lck | Proto-oncogene tyrosine-protein kinase LCK | Src/Lck kinase | T cell-specific protein-tyrosine kinase |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | 57987.83 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P06239 |
|---|
| Residue: | 509 |
|---|
| Sequence: | MGCGCSSHPEDDWMENIDVCENCHYPIVPLDGKGTLLIRNGSEVRDPLVTYEGSNPPASP
LQDNLVIALHSYEPSHDGDLGFEKGEQLRILEQSGEWWKAQSLTTGQEGFIPFNFVAKAN
SLEPEPWFFKNLSRKDAERQLLAPGNTHGSFLIRESESTAGSFSLSVRDFDQNQGEVVKH
YKIRNLDNGGFYISPRITFPGLHELVRHYTNASDGLCTRLSRPCQTQKPQKPWWEDEWEV
PRETLKLVERLGAGQFGEVWMGYYNGHTKVAVKSLKQGSMSPDAFLAEANLMKQLQHQRL
VRLYAVVTQEPIYIITEYMENGSLVDFLKTPSGIKLTINKLLDMAAQIAEGMAFIEERNY
IHRDLRAANILVSDTLSCKIADFGLARLIEDNEYTAREGAKFPIKWTAPEAINYGTFTIK
SDVWSFGILLTEIVTHGRIPYPGMTNPEVIQNLERGYRMVRPDNCPEELYQLMRLCWKER
PEDRPTFDYLRSVLEDFFTATEGQYQPQP
|
|
|
|---|
| BDBM50211223 |
|---|
| n/a |
|---|
| Name | BDBM50211223 |
|---|
| Synonyms: | CHEMBL3939150 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C33H38F3N5O3 |
|---|
| Mol. Mass. | 609.6817 |
|---|
| SMILES | CN1CCN(Cc2ccc(cc2C(F)(F)F)C(=O)Nc2ccc(C)c(OC3CCN(CC3)C(=O)c3ccc(C)nc3)c2)CC1 |
|---|
| Structure | 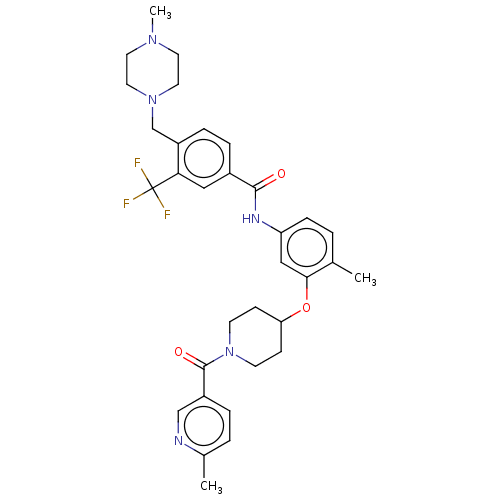
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Wang, Q; Liu, F; Wang, B; Zou, F; Qi, Z; Chen, C; Yu, K; Hu, C; Qi, S; Wang, W; Hu, Z; Liu, J; Wang, W; Wang, L; Liang, Q; Zhang, S; Ren, T; Liu, Q; Liu, J Discovery of 4-Methyl-N-(4-((4-methylpiperazin-1-yl)methyl)-3-(trifluoromethyl)phenyl)-3-((1-nicotinoylpiperidin-4-yl)oxy)benzamide (CHMFL-ABL/KIT-155) as a Novel Highly Potent Type II ABL/KIT Dual Kinase Inhibitor with a Distinct Hinge Binding. J Med Chem60:273-289 (2017) [PubMed] Article
Wang, Q; Liu, F; Wang, B; Zou, F; Qi, Z; Chen, C; Yu, K; Hu, C; Qi, S; Wang, W; Hu, Z; Liu, J; Wang, W; Wang, L; Liang, Q; Zhang, S; Ren, T; Liu, Q; Liu, J Discovery of 4-Methyl-N-(4-((4-methylpiperazin-1-yl)methyl)-3-(trifluoromethyl)phenyl)-3-((1-nicotinoylpiperidin-4-yl)oxy)benzamide (CHMFL-ABL/KIT-155) as a Novel Highly Potent Type II ABL/KIT Dual Kinase Inhibitor with a Distinct Hinge Binding. J Med Chem60:273-289 (2017) [PubMed] Article