| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Glutamate receptor ionotropic, NMDA 1 |
|---|
| Ligand | BDBM50010475 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_140329 |
|---|
| Ki | 8.50±n/a nM |
|---|
| Citation |  Giacobbe, SA; Baraldi, D; Di Fabio, R Unusual synthesis of new glycine antagonists via sequential aldol condensation-lactonization-elimination reaction. Bioorg Med Chem Lett8:1689-92 (1998) [PubMed] Giacobbe, SA; Baraldi, D; Di Fabio, R Unusual synthesis of new glycine antagonists via sequential aldol condensation-lactonization-elimination reaction. Bioorg Med Chem Lett8:1689-92 (1998) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Glutamate receptor ionotropic, NMDA 1 |
|---|
| Name: | Glutamate receptor ionotropic, NMDA 1 |
|---|
| Synonyms: | Glutamate (NMDA) receptor subunit zeta 1 | Glutamate [NMDA] receptor subunit zeta-1 | Glutamate-NMDA-Channel | Glutamate-NMDA-MK801 | Glutamate-NMDA-Polyamine | Grin1 | NMDA | NMDZ1_RAT | Nmdar1 | phencyclidine |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 105533.40 |
|---|
| Organism: | RAT |
|---|
| Description: | P35439 |
|---|
| Residue: | 938 |
|---|
| Sequence: | MSTMHLLTFALLFSCSFARAACDPKIVNIGAVLSTRKHEQMFREAVNQANKRHGSWKIQL
NATSVTHKPNAIQMALSVCEDLISSQVYAILVSHPPTPNDHFTPTPVSYTAGFYRIPVLG
LTTRMSIYSDKSIHLSFLRTVPPYSHQSSVWFEMMRVYNWNHIILLVSDDHEGRAAQKRL
ETLLEERESKAEKVLQFDPGTKNVTALLMEARELEARVIILSASEDDAATVYRAAAMLNM
TGSGYVWLVGEREISGNALRYAPDGIIGLQLINGKNESAHISDAVGVVAQAVHELLEKEN
ITDPPRGCVGNTNIWKTGPLFKRVLMSSKYADGVTGRVEFNEDGDRKFANYSIMNLQNRK
LVQVGIYNGTHVIPNDRKIIWPGGETEKPRGYQMSTRLKIVTIHQEPFVYVKPTMSDGTC
KEEFTVNGDPVKKVICTGPNDTSPGSPRHTVPQCCYGFCIDLLIKLARTMNFTYEVHLVA
DGKFGTQERVNNSNKKEWNGMMGELLSGQADMIVAPLTINNERAQYIEFSKPFKYQGLTI
LVKKEIPRSTLDSFMQPFQSTLWLLVGLSVHVVAVMLYLLDRFSPFGRFKVNSEEEEEDA
LTLSSAMWFSWGVLLNSGIGEGAPRSFSARILGMVWAGFAMIIVASYTANLAAFLVLDRP
EERITGINDPRLRNPSDKFIYATVKQSSVDIYFRRQVELSTMYRHMEKHNYESAAEAIQA
VRDNKLHAFIWDSAVLEFEASQKCDLVTTGELFFRSGFGIGMRKDSPWKQNVSLSILKSH
ENGFMEDLDKTWVRYQECDSRSNAPATLTFENMAGVFMLVAGGIVAGIFLIFIEIAYKRH
KDARRKQMQLAFAAVNVWRKNLQDRKSGRAEPDPKKKATFRAITSTLASSFKRRRSSKDT
STGGGRGALQNQKDTVLPRRAIEREEGQLQLCSRHRES
|
|
|
|---|
| BDBM50010475 |
|---|
| n/a |
|---|
| Name | BDBM50010475 |
|---|
| Synonyms: | GAVESTINEL | GV-150526X |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H12Cl2N2O3 |
|---|
| Mol. Mass. | 375.205 |
|---|
| SMILES | OC(=O)c1[nH]c2cc(Cl)cc(Cl)c2c1\C=C\C(=O)Nc1ccccc1 |
|---|
| Structure | 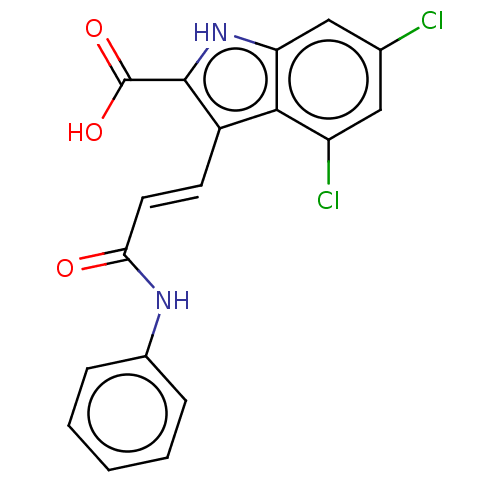
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Giacobbe, SA; Baraldi, D; Di Fabio, R Unusual synthesis of new glycine antagonists via sequential aldol condensation-lactonization-elimination reaction. Bioorg Med Chem Lett8:1689-92 (1998) [PubMed]
Giacobbe, SA; Baraldi, D; Di Fabio, R Unusual synthesis of new glycine antagonists via sequential aldol condensation-lactonization-elimination reaction. Bioorg Med Chem Lett8:1689-92 (1998) [PubMed]