

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Cytochrome P450 3A4 | ||
| Ligand | BDBM50237522 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_1660136 (CHEMBL4009748) | ||
| IC50 | 20000±n/a nM | ||
| Citation |  Wu, YJ; Guernon, J; Shi, J; Ditta, J; Robbins, KJ; Rajamani, R; Easton, A; Newton, A; Bourin, C; Mosure, K; Soars, MG; Knox, RJ; Matchett, M; Pieschl, RL; Post-Munson, DJ; Wang, S; Herrington, J; Graef, J; Newberry, K; Bristow, LJ; Meanwell, NA; Olson, R; Thompson, LA; Dzierba, C Development of New Benzenesulfonamides As Potent and Selective Na J Med Chem60:2513-2525 (2017) [PubMed] Article Wu, YJ; Guernon, J; Shi, J; Ditta, J; Robbins, KJ; Rajamani, R; Easton, A; Newton, A; Bourin, C; Mosure, K; Soars, MG; Knox, RJ; Matchett, M; Pieschl, RL; Post-Munson, DJ; Wang, S; Herrington, J; Graef, J; Newberry, K; Bristow, LJ; Meanwell, NA; Olson, R; Thompson, LA; Dzierba, C Development of New Benzenesulfonamides As Potent and Selective Na J Med Chem60:2513-2525 (2017) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Cytochrome P450 3A4 | |||
| Name: | Cytochrome P450 3A4 | ||
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase | ||
| Type: | Enzyme | ||
| Mol. Mass.: | 57349.57 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | n/a | ||
| Residue: | 503 | ||
| Sequence: |
| ||
| BDBM50237522 | |||
| n/a | |||
| Name | BDBM50237522 | ||
| Synonyms: | CHEMBL4060154 | US10836758, Example 17 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C17H22ClFN4O2S2 | ||
| Mol. Mass. | 432.964 | ||
| SMILES | N[C@H]1CC[C@@H](CCNc2cc(F)c(cc2Cl)S(=O)(=O)Nc2nccs2)CC1 |r,wU:4.4,1.0,(2.3,-59.56,;2.29,-58.02,;.96,-57.25,;.96,-55.71,;2.3,-54.94,;2.3,-53.39,;3.62,-52.63,;3.61,-51.07,;4.95,-50.28,;4.95,-48.74,;6.28,-47.97,;6.28,-46.43,;7.61,-48.73,;7.61,-50.28,;6.27,-51.05,;6.28,-52.59,;8.95,-47.95,;8.18,-46.61,;9.72,-46.61,;10.3,-48.72,;11.63,-47.94,;11.62,-46.41,;13.08,-45.93,;13.99,-47.17,;13.09,-48.42,;3.62,-55.7,;3.62,-57.25,)| | ||
| Structure | 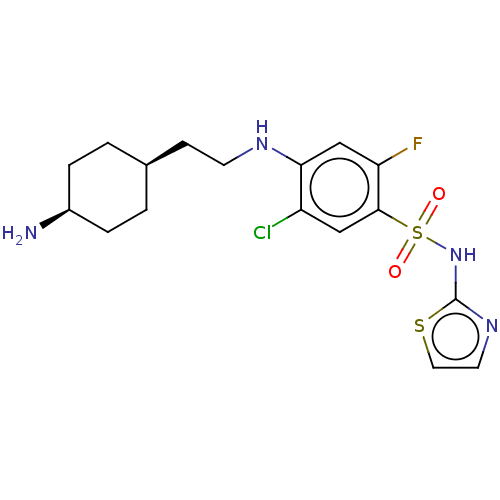
| ||