

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Estrogen receptor | ||
| Ligand | BDBM50238721 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_1664056 (CHEMBL4013737) | ||
| Ki | 1.1±n/a nM | ||
| Citation |  Min, J; Guillen, VS; Sharma, A; Zhao, Y; Ziegler, Y; Gong, P; Mayne, CG; Srinivasan, S; Kim, SH; Carlson, KE; Nettles, KW; Katzenellenbogen, BS; Katzenellenbogen, JA Adamantyl Antiestrogens with Novel Side Chains Reveal a Spectrum of Activities in Suppressing Estrogen Receptor Mediated Activities in Breast Cancer Cells. J Med Chem60:6321-6336 (2017) [PubMed] Article Min, J; Guillen, VS; Sharma, A; Zhao, Y; Ziegler, Y; Gong, P; Mayne, CG; Srinivasan, S; Kim, SH; Carlson, KE; Nettles, KW; Katzenellenbogen, BS; Katzenellenbogen, JA Adamantyl Antiestrogens with Novel Side Chains Reveal a Spectrum of Activities in Suppressing Estrogen Receptor Mediated Activities in Breast Cancer Cells. J Med Chem60:6321-6336 (2017) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Estrogen receptor | |||
| Name: | Estrogen receptor | ||
| Synonyms: | ER | ER-alpha | ESR | ESR1 | ESR1_HUMAN | Estradiol receptor | Estrogen receptor | Estrogen receptor (ER alpha) | Estrogen receptor (ER-alpha) | Estrogen receptor alpha (ER alpha) | Estrogen receptor alpha (ER) | NR3A1 | Nuclear receptor subfamily 3 group A member 1 | ||
| Type: | Protein | ||
| Mol. Mass.: | 66230.44 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P03372 | ||
| Residue: | 595 | ||
| Sequence: |
| ||
| BDBM50238721 | |||
| n/a | |||
| Name | BDBM50238721 | ||
| Synonyms: | CHEMBL4071436 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C29H33NO2 | ||
| Mol. Mass. | 427.5778 | ||
| SMILES | CCCNC(=O)\C=C\c1ccc(cc1)C(=C1C2CC3CC(C2)CC1C3)c1ccc(O)cc1 |TLB:14:15:17:21.19.20,THB:19:18:15:21.20.22,19:20:17.18.24:15,22:20:17:24.23.15,22:23:17:21.19.20,(26.38,-40.99,;27.71,-40.23,;29.04,-41,;30.38,-40.24,;31.71,-41.01,;31.7,-42.55,;33.05,-40.24,;34.38,-41.02,;35.72,-40.25,;35.72,-38.7,;37.05,-37.93,;38.38,-38.7,;38.39,-40.25,;37.05,-41.02,;39.72,-37.92,;41.05,-38.69,;41.06,-40.18,;39.86,-41.45,;41.36,-41.03,;42.77,-41.59,;43.78,-40.31,;42.38,-40.66,;43.8,-38.79,;42.39,-38.21,;41.35,-39.44,;39.71,-36.38,;41.04,-35.61,;41.04,-34.08,;39.7,-33.31,;39.69,-31.77,;38.37,-34.09,;38.38,-35.63,)| | ||
| Structure | 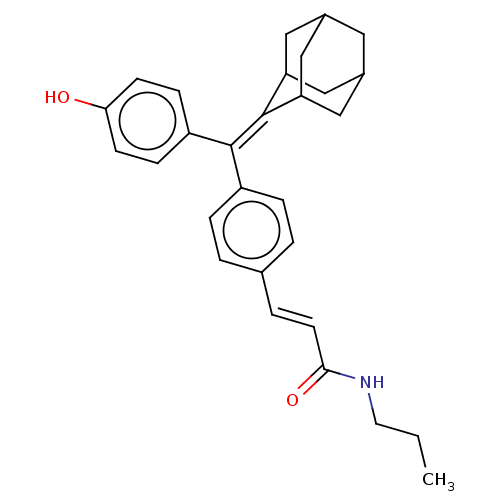
| ||