| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Substance-P receptor |
|---|
| Ligand | BDBM50133817 |
|---|
| Substrate/Competitor | n/a |
|---|
| Ki | >10000±n/a nM |
|---|
| Comments | PDSP_7569 |
|---|
| Citation |  Valenzano, KJ; Grant, ER; Wu, G; Hachicha, M; Schmid, L; Tafesse, L; Sun, Q; Rotshteyn, Y; Francis, J; Limberis, J; Malik, S; Whittemore, ER; Hodges, D N-(4-tertiarybutylphenyl)-4-(3-chloropyridin-2-yl)tetrahydropyrazine -1(2H)-carbox-amide (BCTC), a novel, orally effective vanilloid receptor 1 antagonist with analgesic properties: I. in vitro characterization and pharmacokinetic properties. J Pharmacol Exp Ther306:377-86 (2003) [PubMed] Article Valenzano, KJ; Grant, ER; Wu, G; Hachicha, M; Schmid, L; Tafesse, L; Sun, Q; Rotshteyn, Y; Francis, J; Limberis, J; Malik, S; Whittemore, ER; Hodges, D N-(4-tertiarybutylphenyl)-4-(3-chloropyridin-2-yl)tetrahydropyrazine -1(2H)-carbox-amide (BCTC), a novel, orally effective vanilloid receptor 1 antagonist with analgesic properties: I. in vitro characterization and pharmacokinetic properties. J Pharmacol Exp Ther306:377-86 (2003) [PubMed] Article |
|---|
| More Info.: | Get all data from this article |
|---|
| |
| Substance-P receptor |
|---|
| Name: | Substance-P receptor |
|---|
| Synonyms: | NK-1 receptor | NK-1R | NK1 Receptor | NK1R_RAT | Neurokinin 1 receptor | Neurokinin NK1 | SPR | Substance-P receptor | Tac1r | Tachykinin receptor 1 | Tacr1 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 46371.54 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | Competition binding assays were carried out using membrane preparations from transfected CHO cells that constitutively expressed the rat NK1 receptor. |
|---|
| Residue: | 407 |
|---|
| Sequence: | MDNVLPMDSDLFPNISTNTSESNQFVQPTWQIVLWAAAYTVIVVTSVVGNVVVIWIILAH
KRMRTVTNYFLVNLAFAEACMAAFNTVVNFTYAVHNVWYYGLFYCKFHNFFPIAALFASI
YSMTAVAFDRYMAIIHPLQPRLSATATKVVIFVIWVLALLLAFPQGYYSTTETMPSRVVC
MIEWPEHPNRTYEKAYHICVTVLIYFLPLLVIGYAYTVVGITLWASEIPGDSSDRYHEQV
SAKRKVVKMMIVVVCTFAICWLPFHVFFLLPYINPDLYLKKFIQQVYLASMWLAMSSTMY
NPIIYCCLNDRFRLGFKHAFRCCPFISAGDYEGLEMKSTRYLQTQSSVYKVSRLETTIST
VVGAHEEEPEEGPKATPSSLDLTSNGSSRSNSKTMTESSSFYSNMLA
|
|
|
|---|
| BDBM50133817 |
|---|
| n/a |
|---|
| Name | BDBM50133817 |
|---|
| Synonyms: | 4-(3-Chloro-pyridin-2-yl)-piperazine-1-carboxylic acid (4-tert-butyl-phenyl)-amide | BCTC | CHEMBL441472 | N-(4-tert-butylphenyl)-4-(3-chloropyridin-2-yl)piperazine-1-carboxamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H25ClN4O |
|---|
| Mol. Mass. | 372.892 |
|---|
| SMILES | CC(C)(C)c1ccc(NC(=O)N2CCN(CC2)c2ncccc2Cl)cc1 |
|---|
| Structure | 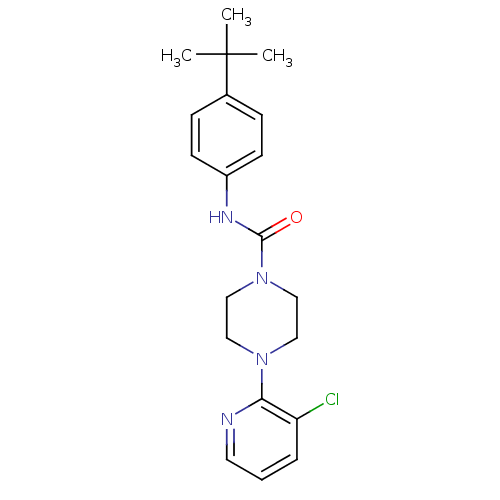
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Valenzano, KJ; Grant, ER; Wu, G; Hachicha, M; Schmid, L; Tafesse, L; Sun, Q; Rotshteyn, Y; Francis, J; Limberis, J; Malik, S; Whittemore, ER; Hodges, D N-(4-tertiarybutylphenyl)-4-(3-chloropyridin-2-yl)tetrahydropyrazine -1(2H)-carbox-amide (BCTC), a novel, orally effective vanilloid receptor 1 antagonist with analgesic properties: I. in vitro characterization and pharmacokinetic properties. J Pharmacol Exp Ther306:377-86 (2003) [PubMed] Article
Valenzano, KJ; Grant, ER; Wu, G; Hachicha, M; Schmid, L; Tafesse, L; Sun, Q; Rotshteyn, Y; Francis, J; Limberis, J; Malik, S; Whittemore, ER; Hodges, D N-(4-tertiarybutylphenyl)-4-(3-chloropyridin-2-yl)tetrahydropyrazine -1(2H)-carbox-amide (BCTC), a novel, orally effective vanilloid receptor 1 antagonist with analgesic properties: I. in vitro characterization and pharmacokinetic properties. J Pharmacol Exp Ther306:377-86 (2003) [PubMed] Article