| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Thromboxane-A synthase |
|---|
| Ligand | BDBM50188619 |
|---|
| Substrate/Competitor | n/a |
|---|
| Ki | 1.1±n/a nM |
|---|
| Comments | PDSP_4501 |
|---|
| Citation |  Hanson, J; Rolin, S; Reynaud, D; Qiao, N; Kelley, LP; Reid, HM; Valentin, F; Tippins, J; Kinsella, BT; Masereel, B; Pace-Asciak, C; Pirotte, B; Dogné, JM In vitro and in vivo pharmacological characterization of BM-613 [N-n-pentyl-N'-[2-(4'-methylphenylamino)-5-nitrobenzenesulfonyl]urea], a novel dual thromboxane synthase inhibitor and thromboxane receptor antagonist. J Pharmacol Exp Ther313:293-301 (2005) [PubMed] Article Hanson, J; Rolin, S; Reynaud, D; Qiao, N; Kelley, LP; Reid, HM; Valentin, F; Tippins, J; Kinsella, BT; Masereel, B; Pace-Asciak, C; Pirotte, B; Dogné, JM In vitro and in vivo pharmacological characterization of BM-613 [N-n-pentyl-N'-[2-(4'-methylphenylamino)-5-nitrobenzenesulfonyl]urea], a novel dual thromboxane synthase inhibitor and thromboxane receptor antagonist. J Pharmacol Exp Ther313:293-301 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article |
|---|
| |
| Thromboxane-A synthase |
|---|
| Name: | Thromboxane-A synthase |
|---|
| Synonyms: | CYP5 | CYP5A1 | Cytochrome P450 5A1 | P450 TxA2 | TBXAS1 | THAS_HUMAN | TXA synthase | TXAS | TXS | Thromboxane A2 Synthase | Thromboxane A2 Synthase (P450 TxA2) | Thromboxane Alpha | Thromboxane prostanoid | Thromboxane synthase | Thromboxane-A synthase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 60524.67 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P24557 |
|---|
| Residue: | 533 |
|---|
| Sequence: | MEALGFLKLEVNGPMVTVALSVALLALLKWYSTSAFSRLEKLGLRHPKPSPFIGNLTFFR
QGFWESQMELRKLYGPLCGYYLGRRMFIVISEPDMIKQVLVENFSNFTNRMASGLEFKSV
ADSVLFLRDKRWEEVRGALMSAFSPEKLNEMVPLISQACDLLLAHLKRYAESGDAFDIQR
CYCNYTTDVVASVAFGTPVDSWQAPEDPFVKHCKRFFEFCIPRPILVLLLSFPSIMVPLA
RILPNKNRDELNGFFNKLIRNVIALRDQQAAEERRRDFLQMVLDARHSASPMGVQDFDIV
RDVFSSTGCKPNPSRQHQPSPMARPLTVDEIVGQAFIFLIAGYEIITNTLSFATYLLATN
PDCQEKLLREVDVFKEKHMAPEFCSLEEGLPYLDMVIAETLRMYPPAFRFTREAAQDCEV
LGQRIPAGAVLEMAVGALHHDPEHWPSPETFNPERFTAEARQQHRPFTYLPFGAGPRSCL
GVRLGLLEVKLTLLHVLHKFRFQACPETQVPLQLESKSALGPKNGVYIKIVSR
|
|
|
|---|
| BDBM50188619 |
|---|
| n/a |
|---|
| Name | BDBM50188619 |
|---|
| Synonyms: | 1-(2-(p-toluidino)-5-nitrophenylsulfonyl)-3-tert-butylurea | BM 573 | BM-573 | CHEMBL210602 | N-tert-butyl-N'-[2-(4-methylphenylamino)-5-nitrobenzenesulfonyl]urea |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H22N4O5S |
|---|
| Mol. Mass. | 406.456 |
|---|
| SMILES | Cc1ccc(Nc2ccc(cc2S(=O)(=O)NC(=O)NC(C)(C)C)[N+]([O-])=O)cc1 |
|---|
| Structure | 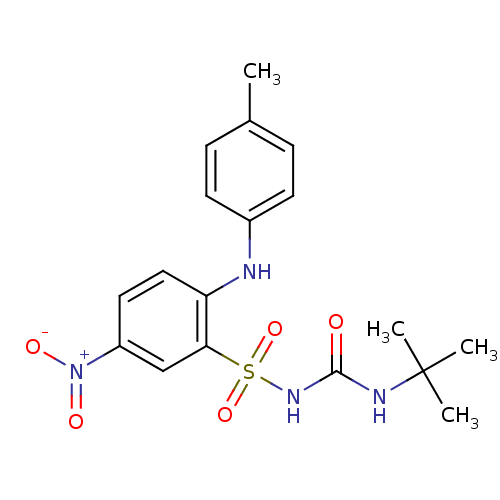
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Hanson, J; Rolin, S; Reynaud, D; Qiao, N; Kelley, LP; Reid, HM; Valentin, F; Tippins, J; Kinsella, BT; Masereel, B; Pace-Asciak, C; Pirotte, B; Dogné, JM In vitro and in vivo pharmacological characterization of BM-613 [N-n-pentyl-N'-[2-(4'-methylphenylamino)-5-nitrobenzenesulfonyl]urea], a novel dual thromboxane synthase inhibitor and thromboxane receptor antagonist. J Pharmacol Exp Ther313:293-301 (2005) [PubMed] Article
Hanson, J; Rolin, S; Reynaud, D; Qiao, N; Kelley, LP; Reid, HM; Valentin, F; Tippins, J; Kinsella, BT; Masereel, B; Pace-Asciak, C; Pirotte, B; Dogné, JM In vitro and in vivo pharmacological characterization of BM-613 [N-n-pentyl-N'-[2-(4'-methylphenylamino)-5-nitrobenzenesulfonyl]urea], a novel dual thromboxane synthase inhibitor and thromboxane receptor antagonist. J Pharmacol Exp Ther313:293-301 (2005) [PubMed] Article