| Reaction Details |
|---|
 | Report a problem with these data |
| Target | High affinity choline transporter 1 |
|---|
| Ligand | BDBM78811 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Dose responses of compounds that activate the Choline Transporter (CHT) - 10 point CRC |
|---|
| EC50 | 22020±n/a nM |
|---|
| Citation |  PubChem, PC Dose responses of compounds that activate the Choline Transporter (CHT) - 10 point CRC PubChem Bioassay(2012)[AID] PubChem, PC Dose responses of compounds that activate the Choline Transporter (CHT) - 10 point CRC PubChem Bioassay(2012)[AID] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| High affinity choline transporter 1 |
|---|
| Name: | High affinity choline transporter 1 |
|---|
| Synonyms: | CHT1 | High-affinity choline transporter | SC5A7_HUMAN | SLC5A7 | high affinity choline transporter 1 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 63194.33 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_1464247 |
|---|
| Residue: | 580 |
|---|
| Sequence: | MAFHVEGLIAIIVFYLLILLVGIWAAWRTKNSGSAEERSEAIIVGGRDIGLLVGGFTMTA
TWVGGGYINGTAEAVYVPGYGLAWAQAPIGYSLSLILGGLFFAKPMRSKGYVTMLDPFQQ
IYGKRMGGLLFIPALMGEMFWAAAIFSALGATISVIIDVDMHISVIISALIATLYTLVGG
LYSVAYTDVVQLFCIFVGLWISVPFALSHPAVADIGFTAVHAKYQKPWLGTVDSSEVYSW
LDSFLLLMLGGIPWQAYFQRVLSSSSATYAQVLSFLAAFGCLVMAIPAILIGAIGASTDW
NQTAYGLPDPKTTEEADMILPIVLQYLCPVYISFFGLGAVSAAVMSSADSSILSASSMFA
RNIYQLSFRQNASDKEIVWVMRITVFVFGASATAMALLTKTVYGLWYLSSDLVYIVIFPQ
LLCVLFVKGTNTYGAVAGYVSGLFLRITGGEPYLYLQPLIFYPGYYPDDNGIYNQKFPFK
TLAMVTSFLTNICISYLAKYLFESGTLPPKLDVFDAVVARHSEENMDKTILVKNENIKLD
ELALVKPRQSMTLSSTFTNKEAFLDVDSSPEGSGTEDNLQ
|
|
|
|---|
| BDBM78811 |
|---|
| n/a |
|---|
| Name | BDBM78811 |
|---|
| Synonyms: | 3-cyclohexyl-N-[2-[(3S,6S)-6-[(4-methoxyphenyl)methyl]-3-(phenylmethyl)-2,3,5,6-tetrahydroimidazo[1,2-a]imidazol-7-yl]ethyl]propanamide | MLS000697877 | N-[2-[(3S,6S)-3-benzyl-6-[(4-methoxyphenyl)methyl]-2,3,5,6-tetrahydroimidazo[1,2-a]imidazol-7-yl]ethyl]-3-cyclohexylpropanamide | N-[2-[(3S,6S)-3-benzyl-6-p-anisyl-2,3,5,6-tetrahydroimidaz[1,2-a]imidazol-7-yl]ethyl]-3-cyclohexyl-propionamide | SMR000386399 | cid_16195624 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C31H42N4O2 |
|---|
| Mol. Mass. | 502.6908 |
|---|
| SMILES | COc1ccc(C[C@H]2CN3[C@@H](Cc4ccccc4)CN=C3N2CCNC(=O)CCC2CCCCC2)cc1 |c:20| |
|---|
| Structure | 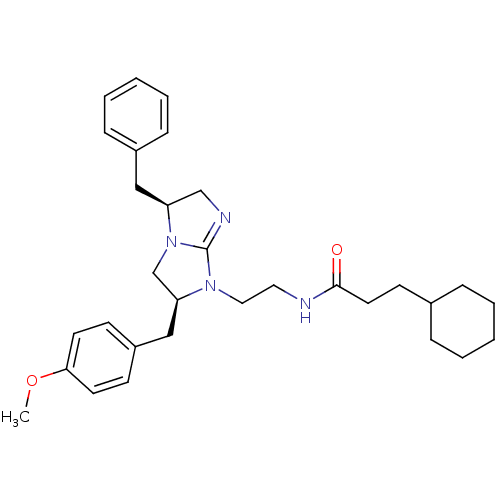
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 PubChem, PC Dose responses of compounds that activate the Choline Transporter (CHT) - 10 point CRC PubChem Bioassay(2012)[AID]
PubChem, PC Dose responses of compounds that activate the Choline Transporter (CHT) - 10 point CRC PubChem Bioassay(2012)[AID]