| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Methionine--tRNA ligase |
|---|
| Ligand | BDBM95811 |
|---|
| Substrate/Competitor | n/a |
|---|
| IC50 | 755.59±n/a nM |
|---|
| Citation |  PubChem, PC Luminescence-based biochemical high throughput dose response assay for inhibitors of Trypanosoma brucei methionyl tRNA synthetase (MetRS) PubChem Bioassay(2013)[AID] PubChem, PC Luminescence-based biochemical high throughput dose response assay for inhibitors of Trypanosoma brucei methionyl tRNA synthetase (MetRS) PubChem Bioassay(2013)[AID] |
|---|
| More Info.: | Get all data from this article |
|---|
| |
| Methionine--tRNA ligase |
|---|
| Name: | Methionine--tRNA ligase |
|---|
| Synonyms: | methionyl-tRNA synthetase |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 86905.45 |
|---|
| Organism: | Trypanosoma brucei brucei strain 927/4 GUTat10.1 |
|---|
| Description: | gi_71746704 |
|---|
| Residue: | 773 |
|---|
| Sequence: | MALKLLSEKANSQALKVLLCSYYVKRPVEVSLSGAYATPILHHPAFKQPIIAPNEMARVI
LFYSVEPTSNNGGAADSSNGDGTASPVAGLTNLTLEHETWLEWEATTFTRAVHPLYTQRR
QTAESLAVFSYLDKKISENDDRCVYSPAVEGKGAADPTDAVSTFFIDCIVWCAVLPALCE
SGVLRDSEKQQLPHLVKWFNTFQKEQKTLIDNAFENLSVQEAADFLRCPRVYKVSAKVEK
VFFVTSPIYYVNAAPHIGHVYSTLITDVIGRYHRVKGERVFALTGTDEHGQKVAEAAKQK
QVSPYDFTAAVAGEFKKCFEQMDYSIDYFIRTTNEQHKAVVKELWTKLEQKGDIYLGRYE
GWYSISDESFLTPQNITDGVDKDGNPCKVSLESGHVVTWVSEENYMFRLSAFRERLLEWY
HANPGCIVPEFRRREVIRAVEKGLPDLSVSRKKETLHNWAIPVPGNPDHCVYVWLDALTN
YLTGSRLRVDESGKEVSLADDFSELERFPADVHVIGKDILKFHAIYWPAFLLSAGLPLPK
KIVAHGWWTKDRKKISKSLGNVFDPVEKAEEFGYDALKYFLLRESGFSDDGDYSDKNMIA
RLNGELADTLGNLVMRCTSAKINVNGEWPSPAAYTEEDESLIQLIKDLPGTADHYYLIPD
IQKAIIAVFDVLRAINAYVTDMAPWKLVKTDPERLRTVLYITLEGVRVTTLLLSPILPRK
SVVIFDMLGVPEVHRKGIENFEFGAVPPGTRLGPAVEGEVLFSKRSTENTKST
|
|
|
|---|
| BDBM95811 |
|---|
| n/a |
|---|
| Name | BDBM95811 |
|---|
| Synonyms: | MLS001211235 | N-(3-Bromo-phenyl)-3-[(2,4-dihydroxy-benzoyl)-hydrazono]-butyramide | N-[(E)-[3-(3-bromoanilino)-3-keto-1-methyl-propylidene]amino]-2,4-dihydroxy-benzamide | N-[(E)-[4-(3-bromoanilino)-4-oxobutan-2-ylidene]amino]-2,4-dihydroxybenzamide | N-[(E)-[4-[(3-bromophenyl)amino]-4-oxidanylidene-butan-2-ylidene]amino]-2,4-bis(oxidanyl)benzamide | SMR000514085 | cid_9592443 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H16BrN3O4 |
|---|
| Mol. Mass. | 406.231 |
|---|
| SMILES | CC(CC(=O)Nc1cccc(Br)c1)=NNC(=O)c1ccc(O)cc1O |w:13.14| |
|---|
| Structure | 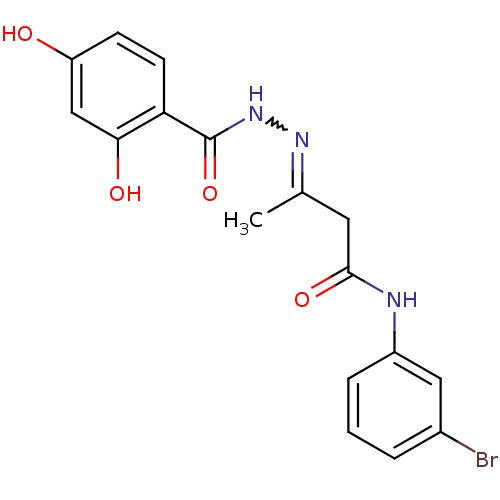
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 PubChem, PC Luminescence-based biochemical high throughput dose response assay for inhibitors of Trypanosoma brucei methionyl tRNA synthetase (MetRS) PubChem Bioassay(2013)[AID]
PubChem, PC Luminescence-based biochemical high throughput dose response assay for inhibitors of Trypanosoma brucei methionyl tRNA synthetase (MetRS) PubChem Bioassay(2013)[AID]