| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Peptidoglycan D,D-transpeptidase FtsI |
|---|
| Ligand | BDBM50240426 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | IC50 Assay |
|---|
| pH | 7.2±n/a |
|---|
| Temperature | 298.15±n/a K |
|---|
| IC50 | 166±62 nM |
|---|
| Comments | extracted |
|---|
| Citation |  van Berkel, SS; Nettleship, JE; Leung, IK; Brem, J; Choi, H; Stuart, DI; Claridge, TD; McDonough, MA; Owens, RJ; Ren, J; Schofield, CJ Binding of (5S)-penicilloic acid to penicillin binding protein 3. ACS Chem Biol8:2112-6 (2013) [PubMed] Article van Berkel, SS; Nettleship, JE; Leung, IK; Brem, J; Choi, H; Stuart, DI; Claridge, TD; McDonough, MA; Owens, RJ; Ren, J; Schofield, CJ Binding of (5S)-penicilloic acid to penicillin binding protein 3. ACS Chem Biol8:2112-6 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Peptidoglycan D,D-transpeptidase FtsI |
|---|
| Name: | Peptidoglycan D,D-transpeptidase FtsI |
|---|
| Synonyms: | Penicillin binding protein 3 (PBP3) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 62820.53 |
|---|
| Organism: | Pseudomonas aeruginosa |
|---|
| Description: | A6VB91 |
|---|
| Residue: | 579 |
|---|
| Sequence: | MKLNHFQGALYPWRFCVIVGLLLAMVGAIVWRIVDLHVIDHDFLKGQGDARSVRHIAIPA
HRGLITDRNGEPLAVSTPVTTLWANPKELMAAKERWPQLAAALGQDTKLFADRIEQNAER
EFIYLVRGLTPEQGEGVISLKVPGVYSIEEFRRFYPAGEVVAHAVGFTDVDDRGREGIEL
AFDEWLAGVPGKRQVLKDRRGRVIKDVQVTKNAKPGKTLALSIDLRLQYLAHRELRNALV
ENGAKAGSLVIMDVKTGEILAMTNQPTYNPNNRRNLQPAAMRNRAMIDVFEPGSTVKPFS
MSAALASGRWKPSDIVDVYPGTLQIGRYTIRDVSRNSRQLDLTGILIKSSNVGISKIAFD
IGAESIYSVMQQVGLGQDTGLGFPGERVGNLPNHRKWPKAETATLAYGYGLSVTAIQLAH
AYAALANDGKSVPLSMTRVDRVPDGVQVISPEVASTVQGMLQQVVEAQGGVFRAQVPGYH
AAGKSGTARKVSVGTKGYRENAYRSLFAGFAPATDPRIAMVVVIDEPSKAGYFGGLVSAP
VFSKVMAGALRLMNVPPDNLPTATEQQQVNAAPAKGGRG
|
|
|
|---|
| BDBM50240426 |
|---|
| n/a |
|---|
| Name | BDBM50240426 |
|---|
| Synonyms: | (2S,5R,6R)-6-{[(2R)-2-{[(4-ethyl-2,3-dioxopiperazin-1-yl)carbonyl]amino}-2-phenylacetyl]amino}-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid | 6beta-{(2R)-2-[(4-ethyl-2,3-dioxopiperazin-1-yl)carboxamido]-2-phenylacetamido}-2,2-dimethylpenam-3alpha-carboxylic acid | CHEMBL702 | Piperacillin | Piperacillin anhydrous |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H27N5O7S |
|---|
| Mol. Mass. | 517.555 |
|---|
| SMILES | CCN1CCN(C(=O)N[C@@H](C(=O)N[C@H]2[C@H]3SC(C)(C)[C@@H](N3C2=O)C(O)=O)c2ccccc2)C(=O)C1=O |r| |
|---|
| Structure | 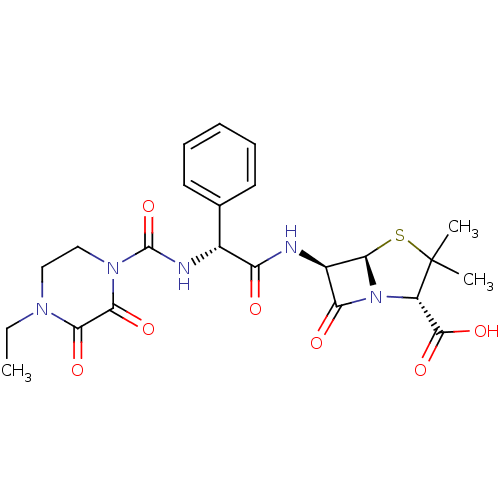
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 van Berkel, SS; Nettleship, JE; Leung, IK; Brem, J; Choi, H; Stuart, DI; Claridge, TD; McDonough, MA; Owens, RJ; Ren, J; Schofield, CJ Binding of (5S)-penicilloic acid to penicillin binding protein 3. ACS Chem Biol8:2112-6 (2013) [PubMed] Article
van Berkel, SS; Nettleship, JE; Leung, IK; Brem, J; Choi, H; Stuart, DI; Claridge, TD; McDonough, MA; Owens, RJ; Ren, J; Schofield, CJ Binding of (5S)-penicilloic acid to penicillin binding protein 3. ACS Chem Biol8:2112-6 (2013) [PubMed] Article