| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Acetylcholinesterase |
|---|
| Ligand | BDBM50089616 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Assay of Substrate Hydrolysis |
|---|
| pH | 7±n/a |
|---|
| Temperature | 298.15±n/a K |
|---|
| Ki | 1.7±0.0 nM |
|---|
| Comments | A- and P-site specific extracted |
|---|
| Citation |  Beri, V; Wildman, SA; Shiomi, K; Al-Rashid, ZF; Cheung, J; Rosenberry, TL The natural product dihydrotanshinone I provides a prototype for uncharged inhibitors that bind specifically to the acetylcholinesterase peripheral site with nanomolar affinity. Biochemistry52:7486-99 (2013) [PubMed] Article Beri, V; Wildman, SA; Shiomi, K; Al-Rashid, ZF; Cheung, J; Rosenberry, TL The natural product dihydrotanshinone I provides a prototype for uncharged inhibitors that bind specifically to the acetylcholinesterase peripheral site with nanomolar affinity. Biochemistry52:7486-99 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Acetylcholinesterase |
|---|
| Name: | Acetylcholinesterase |
|---|
| Synonyms: | ACES_HUMAN | ACHE | Acetylcholinesterase (AChE) | Acetylcholinesterase (human AChE) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 67792.70 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P22303 |
|---|
| Residue: | 614 |
|---|
| Sequence: | MRPPQCLLHTPSLASPLLLLLLWLLGGGVGAEGREDAELLVTVRGGRLRGIRLKTPGGPV
SAFLGIPFAEPPMGPRRFLPPEPKQPWSGVVDATTFQSVCYQYVDTLYPGFEGTEMWNPN
RELSEDCLYLNVWTPYPRPTSPTPVLVWIYGGGFYSGASSLDVYDGRFLVQAERTVLVSM
NYRVGAFGFLALPGSREAPGNVGLLDQRLALQWVQENVAAFGGDPTSVTLFGESAGAASV
GMHLLSPPSRGLFHRAVLQSGAPNGPWATVGMGEARRRATQLAHLVGCPPGGTGGNDTEL
VACLRTRPAQVLVNHEWHVLPQESVFRFSFVPVVDGDFLSDTPEALINAGDFHGLQVLVG
VVKDEGSYFLVYGAPGFSKDNESLISRAEFLAGVRVGVPQVSDLAAEAVVLHYTDWLHPE
DPARLREALSDVVGDHNVVCPVAQLAGRLAAQGARVYAYVFEHRASTLSWPLWMGVPHGY
EIEFIFGIPLDPSRNYTAEEKIFAQRLMRYWANFARTGDPNEPRDPKAPQWPPYTAGAQQ
YVSLDLRPLEVRRGLRAQACAFWNRFLPKLLSATDTLDEAERQWKAEFHRWSSYMVHWKN
QFDHYSKQDRCSDL
|
|
|
|---|
| BDBM50089616 |
|---|
| n/a |
|---|
| Name | BDBM50089616 |
|---|
| Synonyms: | 4a,12a-Dihydroxy-4,4,6a,12b-tetramethyl-9-(3,4,5-trimethoxy-phenyl)-4a,6,6a,12,12a,12b-hexahydro-4H,5H-7,10-dioxa-benzo[a]anthracene-1,11-dione | CHEMBL24686 | territrem B |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C29H34O9 |
|---|
| Mol. Mass. | 526.5749 |
|---|
| SMILES | COc1cc(cc(OC)c1OC)-c1cc2O[C@]3(C)CC[C@@]4(O)C(C)(C)C=CC(=O)[C@]4(C)[C@@]3(O)Cc2c(=O)o1 |r,c:26| |
|---|
| Structure | 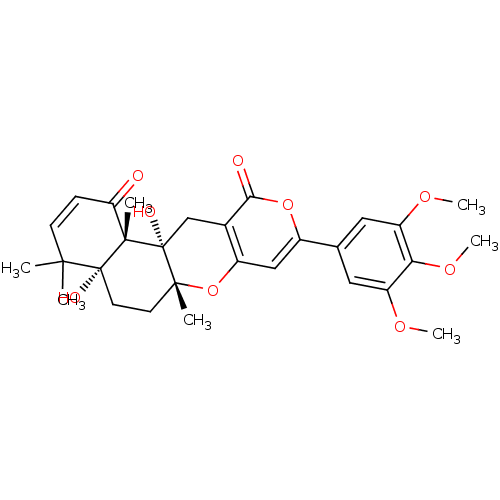
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Beri, V; Wildman, SA; Shiomi, K; Al-Rashid, ZF; Cheung, J; Rosenberry, TL The natural product dihydrotanshinone I provides a prototype for uncharged inhibitors that bind specifically to the acetylcholinesterase peripheral site with nanomolar affinity. Biochemistry52:7486-99 (2013) [PubMed] Article
Beri, V; Wildman, SA; Shiomi, K; Al-Rashid, ZF; Cheung, J; Rosenberry, TL The natural product dihydrotanshinone I provides a prototype for uncharged inhibitors that bind specifically to the acetylcholinesterase peripheral site with nanomolar affinity. Biochemistry52:7486-99 (2013) [PubMed] Article