

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Cyclin-T1 | ||
| Ligand | BDBM126554 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | IMAP Assay | ||
| IC50 | 0.0007±n/a nM | ||
| Citation |  Barsanti, PA; Hu, C; Jin, J; Keyes, R; Kucejko, R; Lin, X; Pan, Y; Pfister, KB; Sendzik, M; Sutton, J; Wan, L Heteroaryl compounds and their uses US Patent US8778951 Publication Date 7/15/2014 Barsanti, PA; Hu, C; Jin, J; Keyes, R; Kucejko, R; Lin, X; Pan, Y; Pfister, KB; Sendzik, M; Sutton, J; Wan, L Heteroaryl compounds and their uses US Patent US8778951 Publication Date 7/15/2014 | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Cyclin-T1 | |||
| Name: | Cyclin-T1 | ||
| Synonyms: | CCNT1 | CCNT1_HUMAN | CycT | CycT1 | Cyclin T | Cyclin T1 | Cyclin-T | ||
| Type: | Enzyme Subunit | ||
| Mol. Mass.: | 80712.40 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | n/a | ||
| Residue: | 726 | ||
| Sequence: |
| ||
| BDBM126554 | |||
| n/a | |||
| Name | BDBM126554 | ||
| Synonyms: | US8778951, 365 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C27H40ClN5O3S | ||
| Mol. Mass. | 550.156 | ||
| SMILES | COC[C@@H](C)N[C@H]1CC[C@@H](CC1)Nc1cc(c(Cl)cn1)-c1cccc(NCC2(C)CCS(=O)(=O)CC2)n1 |r,wU:9.12,wD:6.5,3.3,(8,3.36,;6.67,4.13,;5.33,3.36,;4,4.13,;4,5.67,;2.67,3.36,;1.33,4.13,;1.33,5.67,;,6.44,;-1.33,5.67,;-1.33,4.13,;,3.36,;-2.67,6.44,;-4,5.67,;-4,4.13,;-5.33,3.36,;-6.67,4.13,;-8,3.36,;-6.67,5.67,;-5.33,6.44,;-5.33,1.82,;-6.67,1.05,;-6.67,-.49,;-5.33,-1.26,;-4,-.49,;-2.67,-1.26,;-2.67,-2.8,;-1.33,-3.57,;-1.33,-2.03,;-1.33,-5.11,;,-5.88,;1.33,-5.11,;2.87,-5.11,;2.1,-6.44,;1.33,-3.57,;,-2.8,;-4,1.05,)| | ||
| Structure | 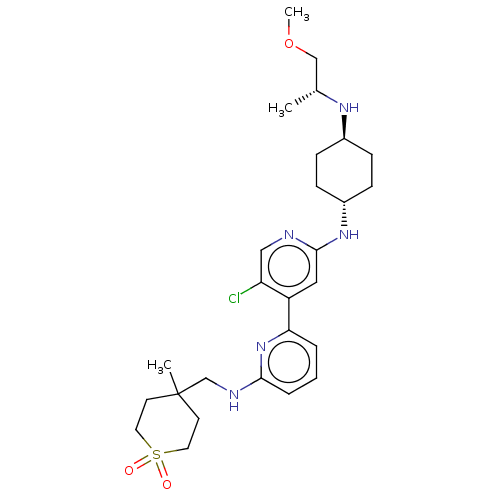
| ||