| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Gag-Pol polyprotein |
|---|
| Ligand | BDBM188497 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Strand Trasnfer Assay |
|---|
| IC50 | 1.7e+2± 3e+1 nM |
|---|
| Citation |  Feng, L; Sharma, A; Slaughter, A; Jena, N; Koh, Y; Shkriabai, N; Larue, RC; Patel, PA; Mitsuya, H; Kessl, JJ; Engelman, A; Fuchs, JR; Kvaratskhelia, M The A128T resistance mutation reveals aberrant protein multimerization as the primary mechanism of action of allosteric HIV-1 integrase inhibitors. J Biol Chem288:15813-20 (2013) [PubMed] Article Feng, L; Sharma, A; Slaughter, A; Jena, N; Koh, Y; Shkriabai, N; Larue, RC; Patel, PA; Mitsuya, H; Kessl, JJ; Engelman, A; Fuchs, JR; Kvaratskhelia, M The A128T resistance mutation reveals aberrant protein multimerization as the primary mechanism of action of allosteric HIV-1 integrase inhibitors. J Biol Chem288:15813-20 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Gag-Pol polyprotein |
|---|
| Name: | Gag-Pol polyprotein |
|---|
| Synonyms: | Capsid protein p24 | Gag-Pol polyprotein (Capsid Protein) | HIV-1 integrase (IN) WT | POL_HV1N5 | Reverse transcriptase (HIV-1 RT) | gag-pol |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 161820.05 |
|---|
| Organism: | Human immunodeficiency virus type 1 group M subtype B (isolate NY5) (HIV-1) |
|---|
| Description: | P12497 |
|---|
| Residue: | 1435 |
|---|
| Sequence: | MGARASVLSGGELDKWEKIRLRPGGKKQYKLKHIVWASRELERFAVNPGLLETSEGCRQI
LGQLQPSLQTGSEELRSLYNTIAVLYCVHQRIDVKDTKEALDKIEEEQNKSKKKAQQAAA
DTGNNSQVSQNYPIVQNLQGQMVHQAISPRTLNAWVKVVEEKAFSPEVIPMFSALSEGAT
PQDLNTMLNTVGGHQAAMQMLKETINEEAAEWDRLHPVHAGPIAPGQMREPRGSDIAGTT
STLQEQIGWMTHNPPIPVGEIYKRWIILGLNKIVRMYSPTSILDIRQGPKEPFRDYVDRF
YKTLRAEQASQEVKNWMTETLLVQNANPDCKTILKALGPGATLEEMMTACQGVGGPGHKA
RVLAEAMSQVTNPATIMIQKGNFRNQRKTVKCFNCGKEGHIAKNCRAPRKKGCWKCGKEG
HQMKDCTERQANFLREDLAFPQGKAREFSSEQTRANSPTRRELQVWGRDNNSLSEAGADR
QGTVSFSFPQITLWQRPLVTIKIGGQLKEALLDTGADDTVLEEMNLPGRWKPKMIGGIGG
FIKVRQYDQILIEICGHKAIGTVLVGPTPVNIIGRNLLTQIGCTLNFPISPIETVPVKLK
PGMDGPKVKQWPLTEEKIKALVEICTEMEKEGKISKIGPENPYNTPVFAIKKKDSTKWRK
LVDFRELNKRTQDFWEVQLGIPHPAGLKQKKSVTVLDVGDAYFSVPLDKDFRKYTAFTIP
SINNETPGIRYQYNVLPQGWKGSPAIFQCSMTKILEPFRKQNPDIVIYQYMDDLYVGSDL
EIGQHRTKIEELRQHLLRWGFTTPDKKHQKEPPFLWMGYELHPDKWTVQPIVLPEKDSWT
VNDIQKLVGKLNWASQIYAGIKVRQLCKLLRGTKALTEVVPLTEEAELELAENREILKEP
VHGVYYDPSKDLIAEIQKQGQGQWTYQIYQEPFKNLKTGKYARMKGAHTNDVKQLTEAVQ
KIATESIVIWGKTPKFKLPIQKETWEAWWTEYWQATWIPEWEFVNTPPLVKLWYQLEKEP
IIGAETFYVDGAANRETKLGKAGYVTDRGRQKVVPLTDTTNQKTELQAIHLALQDSGLEV
NIVTDSQYALGIIQAQPDKSESELVSQIIEQLIKKEKVYLAWVPAHKGIGGNEQVDGLVS
AGIRKVLFLDGIDKAQEEHEKYHSNWRAMASDFNLPPVVAKEIVASCDKCQLKGEAMHGQ
VDCSPGIWQLDCTHLEGKVILVAVHVASGYIEAEVIPAETGQETAYFLLKLAGRWPVKTV
HTDNGSNFTSTTVKAACWWAGIKQEFGIPYNPQSQGVIESMNKELKKIIGQVRDQAEHLK
TAVQMAVFIHNFKRKGGIGGYSAGERIVDIIATDIQTKELQKQITKIQNFRVYYRDSRDP
VWKGPAKLLWKGEGAVVIQDNSDIKVVPRRKAKIIRDYGKQMAGDDCVASRQDED
|
|
|
|---|
| BDBM188497 |
|---|
| n/a |
|---|
| Name | BDBM188497 |
|---|
| Synonyms: | 2-(6-bromo-4-(4-chlorophenyl)-2-methylquinolin-3-yl)-2-(tert-butoxy)acetic acid | ALLINI-2 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H21BrClNO3 |
|---|
| Mol. Mass. | 462.764 |
|---|
| SMILES | Cc1nc2ccc(Br)cc2c(-c2ccc(Cl)cc2)c1C(OC(C)(C)C)C(O)=O |
|---|
| Structure | 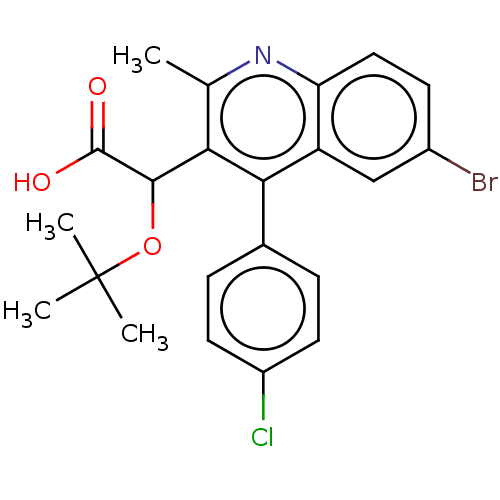
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Feng, L; Sharma, A; Slaughter, A; Jena, N; Koh, Y; Shkriabai, N; Larue, RC; Patel, PA; Mitsuya, H; Kessl, JJ; Engelman, A; Fuchs, JR; Kvaratskhelia, M The A128T resistance mutation reveals aberrant protein multimerization as the primary mechanism of action of allosteric HIV-1 integrase inhibitors. J Biol Chem288:15813-20 (2013) [PubMed] Article
Feng, L; Sharma, A; Slaughter, A; Jena, N; Koh, Y; Shkriabai, N; Larue, RC; Patel, PA; Mitsuya, H; Kessl, JJ; Engelman, A; Fuchs, JR; Kvaratskhelia, M The A128T resistance mutation reveals aberrant protein multimerization as the primary mechanism of action of allosteric HIV-1 integrase inhibitors. J Biol Chem288:15813-20 (2013) [PubMed] Article