| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Lipoxygenase |
|---|
| Ligand | BDBM22334 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Colorimetric 5-LO Assay |
|---|
| IC50 | 1.71e+4±n/a nM |
|---|
| Citation |  Richardson-Sanchez, T; Tieu, W; Codd, R Reverse Biosynthesis: Generating Combinatorial Pools of Drug Leads from Enzyme-Mediated Fragmentation of Natural Products. Chembiochem18:368-373 (2017) [PubMed] Article Richardson-Sanchez, T; Tieu, W; Codd, R Reverse Biosynthesis: Generating Combinatorial Pools of Drug Leads from Enzyme-Mediated Fragmentation of Natural Products. Chembiochem18:368-373 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Lipoxygenase |
|---|
| Name: | Lipoxygenase |
|---|
| Synonyms: | 5-Lipoxygenase | 5-Lipoxygenase (5-LO) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 97294.59 |
|---|
| Organism: | Solanum tuberosum (potato) |
|---|
| Description: | n/a |
|---|
| Residue: | 864 |
|---|
| Sequence: | MNIGQIMGGRELFGGHDDSKKVKGTVVMMKKNALDFTDLAGSLTDIAFDVLGQKVSFQLI
SSVQGDPTNGLQGKHSNPAYLENSLFTLTPLTAGSETAFGVTFDWNEEFGVPGAFIIKNT
HINEFFLKSLTLEDVPNHGKVHFDCNSWVYPSFRYKSDRIFFANQPYLPSKTPELLRKYR
ENELLTLRGDGTGKREAWDRIYDYDIYNDLGNPDQGKENVRTTLGGSAEYPYPRRGRTGR
PPTRTDPKSESRIPLLLSLDIYVPRDERFGHLKMSDFLTYALKSIVQFILPELHALFDGT
PNEFDSFEDVLRLYEGGIKLPQGPLFKALTAAIPLEMIKELLRTDGEGILRFPTPLVIKD
SKTAWRTDEEFAREMLAGVNPIIISRLQEFPPKSKLDPEAYGNQNSTITAEHIEDKLDGL
TVDEAMNNNKLFILNHHDLLIPYLRRINTTITKSYASRTLLFLQDNGSLKPLAIELSLPH
PDGDQFGVTSKVYTPSDQGVESSIWQLAKAYVAVNDAGVHQLISHWLNTHAVIEPFVIAT
NRQLSVLHPIHKLLYPHFRDTMNINASARQILINAGGVLESTVFQSKFALEMSAVVYKDW
VFPDQALPADLVKRGVAVEDSSSPHGVRLLIEDYPYAVDGLEIWSAIKSWVTDYCSFYYG
SDEEILKDNELQAWWKELREVGHGDKKNEPWWPEMETPQELIDSCTTIIWIASALHAAVN
FGQYPYAGYLPNRPTVSRRFMPEPGTPEYEELKKNPDKAFLKTITAQLQTLLGVSLVEIL
SRHTTDEIYLGQRESPEWTKDKEPLAAFDKFGKKLTDIEKQIIQRNGDNILTNRSGPVNA
PYTLLFPTSEGGLTGKGIPNSVSI
|
|
|
|---|
| BDBM22334 |
|---|
| n/a |
|---|
| Name | BDBM22334 |
|---|
| Synonyms: | BW A4C | BW4C | BWA4C | BWA4C, 10 | CHEMBL314360 | JMC515449 Compound 7 | N-(3-phenoxycinnamyl)acetohydroxamic acid | N-hydroxy-N-[(2E)-3-(3-phenoxyphenyl)prop-2-en-1-yl]acetamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H17NO3 |
|---|
| Mol. Mass. | 283.3218 |
|---|
| SMILES | CC(=O)N(O)C\C=C\c1cccc(Oc2ccccc2)c1 |
|---|
| Structure | 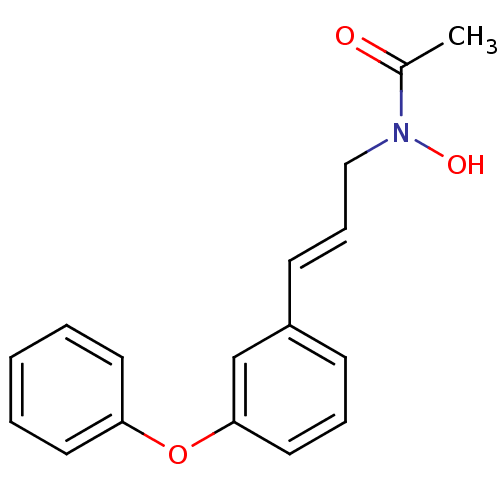
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Richardson-Sanchez, T; Tieu, W; Codd, R Reverse Biosynthesis: Generating Combinatorial Pools of Drug Leads from Enzyme-Mediated Fragmentation of Natural Products. Chembiochem18:368-373 (2017) [PubMed] Article
Richardson-Sanchez, T; Tieu, W; Codd, R Reverse Biosynthesis: Generating Combinatorial Pools of Drug Leads from Enzyme-Mediated Fragmentation of Natural Products. Chembiochem18:368-373 (2017) [PubMed] Article