| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Polycomb protein EED |
|---|
| Ligand | BDBM223987 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Radioligand Binding Assay |
|---|
| Temperature | 296.15±0 K |
|---|
| IC50 | 18±2 nM |
|---|
| Citation |  He, Y; Selvaraju, S; Curtin, ML; Jakob, CG; Zhu, H; Comess, KM; Shaw, B; The, J; Lima-Fernandes, E; Szewczyk, MM; Cheng, D; Klinge, KL; Li, HQ; Pliushchev, M; Algire, MA; Maag, D; Guo, J; Dietrich, J; Panchal, SC; Petros, AM; Sweis, RF; Torrent, M; Bigelow, LJ; Senisterra, G; Li, F; Kennedy, S; Wu, Q; Osterling, DJ; Lindley, DJ; Gao, W; Galasinski, S; Barsyte-Lovejoy, D; Vedadi, M; Buchanan, FG; Arrowsmith, CH; Chiang, GG; Sun, C; Pappano, WN The EED protein-protein interaction inhibitor A-395 inactivates the PRC2 complex. Nat Chem Biol13:389-395 (2017) [PubMed] Article He, Y; Selvaraju, S; Curtin, ML; Jakob, CG; Zhu, H; Comess, KM; Shaw, B; The, J; Lima-Fernandes, E; Szewczyk, MM; Cheng, D; Klinge, KL; Li, HQ; Pliushchev, M; Algire, MA; Maag, D; Guo, J; Dietrich, J; Panchal, SC; Petros, AM; Sweis, RF; Torrent, M; Bigelow, LJ; Senisterra, G; Li, F; Kennedy, S; Wu, Q; Osterling, DJ; Lindley, DJ; Gao, W; Galasinski, S; Barsyte-Lovejoy, D; Vedadi, M; Buchanan, FG; Arrowsmith, CH; Chiang, GG; Sun, C; Pappano, WN The EED protein-protein interaction inhibitor A-395 inactivates the PRC2 complex. Nat Chem Biol13:389-395 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Polycomb protein EED |
|---|
| Name: | Polycomb protein EED |
|---|
| Synonyms: | EED | EED_HUMAN | WAIT-1 | WD protein associating with integrin cytoplasmic tails 1 | hEED |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 50201.68 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | O75530 |
|---|
| Residue: | 441 |
|---|
| Sequence: | MSEREVSTAPAGTDMPAAKKQKLSSDENSNPDLSGDENDDAVSIESGTNTERPDTPTNTP
NAPGRKSWGKGKWKSKKCKYSFKCVNSLKEDHNQPLFGVQFNWHSKEGDPLVFATVGSNR
VTLYECHSQGEIRLLQSYVDADADENFYTCAWTYDSNTSHPLLAVAGSRGIIRIINPITM
QCIKHYVGHGNAINELKFHPRDPNLLLSVSKDHALRLWNIQTDTLVAIFGGVEGHRDEVL
SADYDLLGEKIMSCGMDHSLKLWRINSKRMMNAIKESYDYNPNKTNRPFISQKIHFPDFS
TRDIHRNYVDCVRWLGDLILSKSCENAIVCWKPGKMEDDIDKIKPSESNVTILGRFDYSQ
CDIWYMRFSMDFWQKMLALGNQVGKLYVWDLEVEDPHKAKCTTLTHHKCGAAIRQTSFSR
DSSILIAVCDDASIWRWDRLR
|
|
|
|---|
| BDBM223987 |
|---|
| n/a |
|---|
| Name | BDBM223987 |
|---|
| Synonyms: | A-395 (5) | rac-(3R,4S)-1-(7-fluoro-2,3-dihydro-1H-inden-1-yl)-N,N-dimethyl-4-(4-(4-(methylsulfonyl)piperazin-1-yl)phenyl)pyrrolidin-3-amine (4) |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H35FN4O2S |
|---|
| Mol. Mass. | 486.645 |
|---|
| SMILES | CN(C)[C@H]1CN(C[C@@H]1c1ccc(cc1)N1CCN(CC1)S(C)(=O)=O)C1CCc2cccc(F)c12 |r,w:24.26| |
|---|
| Structure | 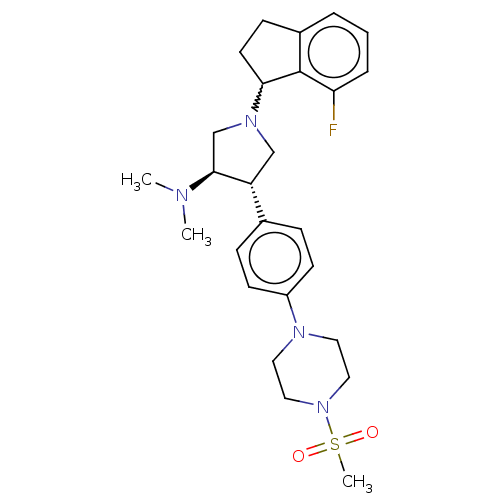
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 He, Y; Selvaraju, S; Curtin, ML; Jakob, CG; Zhu, H; Comess, KM; Shaw, B; The, J; Lima-Fernandes, E; Szewczyk, MM; Cheng, D; Klinge, KL; Li, HQ; Pliushchev, M; Algire, MA; Maag, D; Guo, J; Dietrich, J; Panchal, SC; Petros, AM; Sweis, RF; Torrent, M; Bigelow, LJ; Senisterra, G; Li, F; Kennedy, S; Wu, Q; Osterling, DJ; Lindley, DJ; Gao, W; Galasinski, S; Barsyte-Lovejoy, D; Vedadi, M; Buchanan, FG; Arrowsmith, CH; Chiang, GG; Sun, C; Pappano, WN The EED protein-protein interaction inhibitor A-395 inactivates the PRC2 complex. Nat Chem Biol13:389-395 (2017) [PubMed] Article
He, Y; Selvaraju, S; Curtin, ML; Jakob, CG; Zhu, H; Comess, KM; Shaw, B; The, J; Lima-Fernandes, E; Szewczyk, MM; Cheng, D; Klinge, KL; Li, HQ; Pliushchev, M; Algire, MA; Maag, D; Guo, J; Dietrich, J; Panchal, SC; Petros, AM; Sweis, RF; Torrent, M; Bigelow, LJ; Senisterra, G; Li, F; Kennedy, S; Wu, Q; Osterling, DJ; Lindley, DJ; Gao, W; Galasinski, S; Barsyte-Lovejoy, D; Vedadi, M; Buchanan, FG; Arrowsmith, CH; Chiang, GG; Sun, C; Pappano, WN The EED protein-protein interaction inhibitor A-395 inactivates the PRC2 complex. Nat Chem Biol13:389-395 (2017) [PubMed] Article