| Reaction Details |
|---|
 | Report a problem with these data |
| Target | UDP-glucuronosyltransferase 1-6 |
|---|
| Ligand | BDBM21363 |
|---|
| Substrate/Competitor | 1-Naphthol |
|---|
| Meas. Tech. | UDP-glucuronosyltransferase Activity Assay |
|---|
| IC50 | 2.3e+4± 1e+3 nM |
|---|
| Citation |  Liu, Y; She, M; Wu, Z; Dai, R The inhibition study of human UDP-glucuronosyltransferases with cytochrome P450 selective substrates and inhibitors. J Enzyme Inhib Med Chem26:386-93 (2011) [PubMed] Article Liu, Y; She, M; Wu, Z; Dai, R The inhibition study of human UDP-glucuronosyltransferases with cytochrome P450 selective substrates and inhibitors. J Enzyme Inhib Med Chem26:386-93 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| UDP-glucuronosyltransferase 1-6 |
|---|
| Name: | UDP-glucuronosyltransferase 1-6 |
|---|
| Synonyms: | GNT1 | Phenol-metabolizing UDP-glucuronosyltransferase | UD16_HUMAN | UDP-glucuronosyltransferase 1-6 | UDP-glucuronosyltransferase 1-F | UDP-glucuronosyltransferase 1A6 | UDPGT 1-6 | UGT-1F | UGT1 | UGT1*6 | UGT1-06 | UGT1.6 | UGT1A6 | UGT1F | Uridine-5'-diphosphoglucuronosyltransferase 1A6 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 60765.14 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P19224 |
|---|
| Residue: | 532 |
|---|
| Sequence: | MACLLRSFQRISAGVFFLALWGMVVGDKLLVVPQDGSHWLSMKDIVEVLSDRGHEIVVVV
PEVNLLLKESKYYTRKIYPVPYDQEELKNRYQSFGNNHFAERSFLTAPQTEYRNNMIVIG
LYFINCQSLLQDRDTLNFFKESKFDALFTDPALPCGVILAEYLGLPSVYLFRGFPCSLEH
TFSRSPDPVSYIPRCYTKFSDHMTFSQRVANFLVNLLEPYLFYCLFSKYEELASAVLKRD
VDIITLYQKVSVWLLRYDFVLEYPRPVMPNMVFIGGINCKKRKDLSQEFEAYINASGEHG
IVVFSLGSMVSEIPEKKAMAIADALGKIPQTVLWRYTGTRPSNLANNTILVKWLPQNDLL
GHPMTRAFITHAGSHGVYESICNGVPMVMMPLFGDQMDNAKRMETKGAGVTLNVLEMTSE
DLENALKAVINDKSYKENIMRLSSLHKDRPVEPLDLAVFWVEFVMRHKGAPHLRPAAHDL
TWYQYHSLDVIGFLLAVVLTVAFITFKCCAYGYRKCLGKKGRVKKAHKSKTH
|
|
|
|---|
| BDBM21363 |
|---|
| 1-Naphthol |
|---|
| Name | BDBM21363 |
|---|
| Synonyms: | 12-chloro-9-(2-fluorophenyl)-3-methyl-2,4,8-triazatricyclo[8.4.0.0^{2,6}]tetradeca-1(10),3,5,8,11,13-hexaene | 8-chloro-6-(2-fluorophenyl)-1-methyl-4H-imidazo[1,5-a][1,4]benzodiazepine | CHEMBL655 | Dormicum | Midazolam | Ro 21-3981 | US20230416258, Compound Midazolam |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H13ClFN3 |
|---|
| Mol. Mass. | 325.767 |
|---|
| SMILES | Cc1ncc2CN=C(c3ccccc3F)c3cc(Cl)ccc3-n12 |t:6| |
|---|
| Structure | 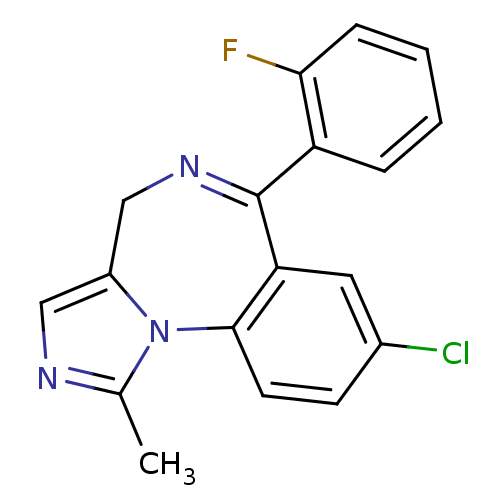
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Liu, Y; She, M; Wu, Z; Dai, R The inhibition study of human UDP-glucuronosyltransferases with cytochrome P450 selective substrates and inhibitors. J Enzyme Inhib Med Chem26:386-93 (2011) [PubMed] Article
Liu, Y; She, M; Wu, Z; Dai, R The inhibition study of human UDP-glucuronosyltransferases with cytochrome P450 selective substrates and inhibitors. J Enzyme Inhib Med Chem26:386-93 (2011) [PubMed] Article