

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Cytochrome P450 2C8 | ||
| Ligand | BDBM253992 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | Inhibition Assays | ||
| IC50 | 900±n/a nM | ||
| Citation |  Reiser, U 6-alkynyl-pyridine derivatives US Patent US9481673 Publication Date 11/1/2016 Reiser, U 6-alkynyl-pyridine derivatives US Patent US9481673 Publication Date 11/1/2016 | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Cytochrome P450 2C8 | |||
| Name: | Cytochrome P450 2C8 | ||
| Synonyms: | CP2C8_HUMAN | CYP2C8 | CYPIIC8 | Cytochrome P450 2C8 (CYP2C8) | P450 IIC2 | P450 MP-12/MP-20 | P450 form 1 | S-mephenytoin 4-hydroxylase | ||
| Type: | Protein | ||
| Mol. Mass.: | 55839.23 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P10632 | ||
| Residue: | 490 | ||
| Sequence: |
| ||
| BDBM253992 | |||
| n/a | |||
| Name | BDBM253992 | ||
| Synonyms: | US9481673, (S)-2 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C35H31N7O | ||
| Mol. Mass. | 565.6669 | ||
| SMILES | CN[C@@H](C)C(=O)Nc1ccc(-c2c(nc3cc(C)ccn23)-c2ccnc(C)c2)c(n1)C#Cc1ccc2c(C)nccc2c1 |r,wU:2.2,(-6,-3.76,;-7.34,-2.99,;-7.34,-1.45,;-8.67,-.68,;-6,-.68,;-6,.86,;-4.67,-1.45,;-3.33,-.68,;-3.33,.86,;-2,1.63,;-.67,.86,;.67,1.63,;.67,3.17,;2.13,3.65,;3.04,2.4,;4.57,2.24,;5.19,.83,;6.68,.44,;4.29,-.41,;2.76,-.25,;2.13,1.16,;-.42,4.26,;-.02,5.75,;-1.11,6.84,;-2.6,6.44,;-3,4.95,;-4.49,4.55,;-1.91,3.86,;-.67,-.68,;-2,-1.45,;.67,-1.45,;2,-2.22,;3.33,-2.99,;3.33,-4.53,;4.67,-5.3,;6,-4.53,;7.34,-5.3,;7.34,-6.84,;8.67,-4.53,;8.67,-2.99,;7.34,-2.22,;6,-2.99,;4.67,-2.22,)| | ||
| Structure | 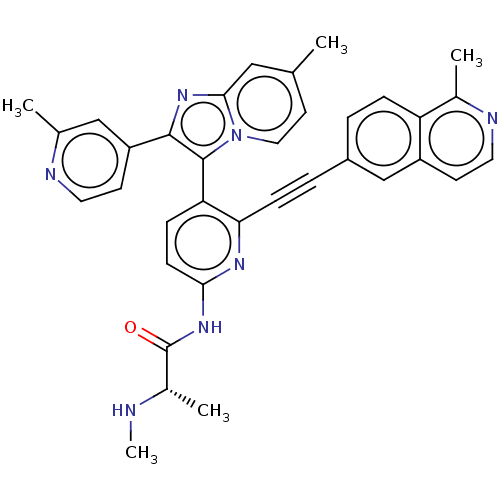
| ||