

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50323699 2-(benzyloxy)naphthalene::2-Benzyloxy-naphthalene::CHEMBL146439
SMILES: C(Oc1ccc2ccccc2c1)c1ccccc1
InChI Key: InChIKey=WLTCCDHHWYAMCG-UHFFFAOYSA-N
Data: 3 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Arachidonate 5-lipoxygenase (Homo sapiens (Human)) | BDBM50323699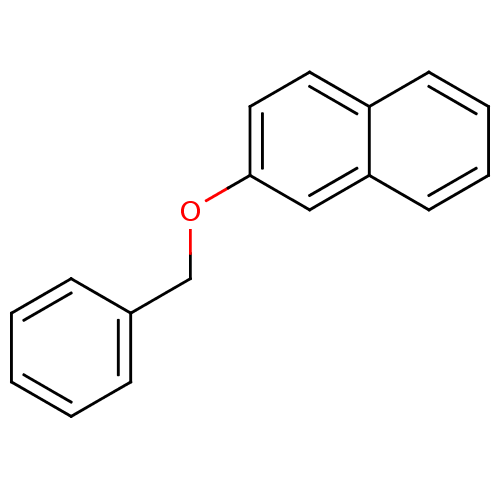 (2-(benzyloxy)naphthalene | 2-Benzyloxy-naphthalene...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 7.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Max Planck Institute of Molecular Physiology Curated by ChEMBL | Assay Description Inhibition of 5-LOX | J Med Chem 55: 5989-6001 (2012) Article DOI: 10.1021/jm300288g BindingDB Entry DOI: 10.7270/Q2J38TPJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Monoamine Oxidase Type B (MAO-B) (Rattus norvegicus (rat)) | BDBM50323699 (2-(benzyloxy)naphthalene | 2-Benzyloxy-naphthalene...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 2.57E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lausanne Curated by ChEMBL | Assay Description Inhibitory effect on Monoamine oxidase B, SD on IC50 values < 10% | J Med Chem 43: 4747-58 (2000) BindingDB Entry DOI: 10.7270/Q20K29RT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Arachidonate 5-lipoxygenase (Homo sapiens (Human)) | BDBM50323699 (2-(benzyloxy)naphthalene | 2-Benzyloxy-naphthalene...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 9.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institut für Molekulare Physiologie Curated by ChEMBL | Assay Description Inhibition of LOX5 in human whole blood assessed as inhibition of conversion of aracidonic acid to leukotrine | Nat Chem Biol 5: 585-92 (2009) Article DOI: 10.1038/nchembio.188 BindingDB Entry DOI: 10.7270/Q2MP53F2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||