 Found 7 hits for monomerid = 245492
Found 7 hits for monomerid = 245492 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Serine-protein kinase ATM
(Homo sapiens (Human)) | BDBM245492
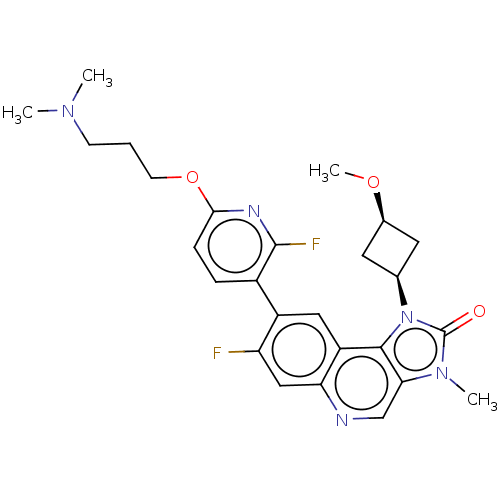
(US9428503, 19)Show SMILES CO[C@H]1C[C@H](C1)n1c2c(cnc3cc(F)c(cc23)-c2ccc(OCCCN(C)C)nc2F)n(C)c1=O |r,wD:4.6,2.1,(3.02,4.9,;2.62,3.41,;3.71,2.32,;3.71,.78,;5.25,.78,;5.25,2.32,;6.34,-.31,;6.02,-1.82,;7.35,-2.59,;7.35,-4.13,;6.02,-4.9,;4.69,-4.13,;3.35,-4.9,;2.02,-4.13,;.68,-4.9,;2.02,-2.59,;3.35,-1.82,;4.69,-2.59,;.68,-1.82,;.68,-.28,;-.65,.49,;-1.98,-.28,;-3.32,.49,;-4.65,-.28,;-5.98,.49,;-7.32,-.28,;-8.65,.49,;-9.98,-.28,;-8.65,2.03,;-1.98,-1.82,;-.65,-2.59,;-.65,-4.13,;8.5,-1.56,;9.98,-1.95,;7.87,-.15,;8.64,1.19,)| Show InChI InChI=1S/C26H29F2N5O3/c1-31(2)8-5-9-36-23-7-6-17(25(28)30-23)18-12-19-21(13-20(18)27)29-14-22-24(19)33(26(34)32(22)3)15-10-16(11-15)35-4/h6-7,12-16H,5,8-11H2,1-4H3/t15-,16+ | PDB
UniProtKB/SwissProt
UniProtKB/TrEMBL
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 2.62 | n/a | n/a | n/a | n/a | n/a | 37 |
AstraZeneca AB
US Patent
| Assay Description
Assay a) HT29 cells (ECACC #85061109) were seeded into 384 well assay plates (Costar #3712) at a density of 3500 cells/well in 40 μl EMEM medium c... |
US Patent US9428503 (2016)
BindingDB Entry DOI: 10.7270/Q2GH9GVJ |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase ATR
(Homo sapiens (Human)) | BDBM245492

(US9428503, 19)Show SMILES CO[C@H]1C[C@H](C1)n1c2c(cnc3cc(F)c(cc23)-c2ccc(OCCCN(C)C)nc2F)n(C)c1=O |r,wD:4.6,2.1,(3.02,4.9,;2.62,3.41,;3.71,2.32,;3.71,.78,;5.25,.78,;5.25,2.32,;6.34,-.31,;6.02,-1.82,;7.35,-2.59,;7.35,-4.13,;6.02,-4.9,;4.69,-4.13,;3.35,-4.9,;2.02,-4.13,;.68,-4.9,;2.02,-2.59,;3.35,-1.82,;4.69,-2.59,;.68,-1.82,;.68,-.28,;-.65,.49,;-1.98,-.28,;-3.32,.49,;-4.65,-.28,;-5.98,.49,;-7.32,-.28,;-8.65,.49,;-9.98,-.28,;-8.65,2.03,;-1.98,-1.82,;-.65,-2.59,;-.65,-4.13,;8.5,-1.56,;9.98,-1.95,;7.87,-.15,;8.64,1.19,)| Show InChI InChI=1S/C26H29F2N5O3/c1-31(2)8-5-9-36-23-7-6-17(25(28)30-23)18-12-19-21(13-20(18)27)29-14-22-24(19)33(26(34)32(22)3)15-10-16(11-15)35-4/h6-7,12-16H,5,8-11H2,1-4H3/t15-,16+ | PDB
Reactome pathway
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | 37 |
AstraZeneca AB
US Patent
| Assay Description
Assay b) HT29 cells (ECACC #85061109) were seeded into 384 well assay plates (Costar #3712) at a density of 6000 cells/well in 40 μl EMEM medium c... |
US Patent US9428503 (2016)
BindingDB Entry DOI: 10.7270/Q2GH9GVJ |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM245492

(US9428503, 19)Show SMILES CO[C@H]1C[C@H](C1)n1c2c(cnc3cc(F)c(cc23)-c2ccc(OCCCN(C)C)nc2F)n(C)c1=O |r,wD:4.6,2.1,(3.02,4.9,;2.62,3.41,;3.71,2.32,;3.71,.78,;5.25,.78,;5.25,2.32,;6.34,-.31,;6.02,-1.82,;7.35,-2.59,;7.35,-4.13,;6.02,-4.9,;4.69,-4.13,;3.35,-4.9,;2.02,-4.13,;.68,-4.9,;2.02,-2.59,;3.35,-1.82,;4.69,-2.59,;.68,-1.82,;.68,-.28,;-.65,.49,;-1.98,-.28,;-3.32,.49,;-4.65,-.28,;-5.98,.49,;-7.32,-.28,;-8.65,.49,;-9.98,-.28,;-8.65,2.03,;-1.98,-1.82,;-.65,-2.59,;-.65,-4.13,;8.5,-1.56,;9.98,-1.95,;7.87,-.15,;8.64,1.19,)| Show InChI InChI=1S/C26H29F2N5O3/c1-31(2)8-5-9-36-23-7-6-17(25(28)30-23)18-12-19-21(13-20(18)27)29-14-22-24(19)33(26(34)32(22)3)15-10-16(11-15)35-4/h6-7,12-16H,5,8-11H2,1-4H3/t15-,16+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | >2.80E+4 | n/a | n/a | n/a | n/a | n/a | 37 |
AstraZeneca AB
US Patent
| Assay Description
Assay c) BT474 cells (human breast ductal carcinoma, ATCC HTB-20) were seeded into black 384 well plates (Costar, #3712) at a density of 5600 cells/w... |
US Patent US9428503 (2016)
BindingDB Entry DOI: 10.7270/Q2GH9GVJ |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase ATR
(Homo sapiens (Human)) | BDBM245492

(US9428503, 19)Show SMILES CO[C@H]1C[C@H](C1)n1c2c(cnc3cc(F)c(cc23)-c2ccc(OCCCN(C)C)nc2F)n(C)c1=O |r,wD:4.6,2.1,(3.02,4.9,;2.62,3.41,;3.71,2.32,;3.71,.78,;5.25,.78,;5.25,2.32,;6.34,-.31,;6.02,-1.82,;7.35,-2.59,;7.35,-4.13,;6.02,-4.9,;4.69,-4.13,;3.35,-4.9,;2.02,-4.13,;.68,-4.9,;2.02,-2.59,;3.35,-1.82,;4.69,-2.59,;.68,-1.82,;.68,-.28,;-.65,.49,;-1.98,-.28,;-3.32,.49,;-4.65,-.28,;-5.98,.49,;-7.32,-.28,;-8.65,.49,;-9.98,-.28,;-8.65,2.03,;-1.98,-1.82,;-.65,-2.59,;-.65,-4.13,;8.5,-1.56,;9.98,-1.95,;7.87,-.15,;8.64,1.19,)| Show InChI InChI=1S/C26H29F2N5O3/c1-31(2)8-5-9-36-23-7-6-17(25(28)30-23)18-12-19-21(13-20(18)27)29-14-22-24(19)33(26(34)32(22)3)15-10-16(11-15)35-4/h6-7,12-16H,5,8-11H2,1-4H3/t15-,16+ | PDB
Reactome pathway
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of ATR in human HT29 cells assessed as decrease in Chk1 phosphorylation at Ser 345 preincubated for 1 hr followed by 4NQO addition and mea... |
J Med Chem 61: 3823-3841 (2018)
Article DOI: 10.1021/acs.jmedchem.7b01896
BindingDB Entry DOI: 10.7270/Q2RB775P |
More data for this
Ligand-Target Pair | |
1,3-beta-glucan synthase component GLS2
(Saccharomyces cerevisiae) | BDBM245492

(US9428503, 19)Show SMILES CO[C@H]1C[C@H](C1)n1c2c(cnc3cc(F)c(cc23)-c2ccc(OCCCN(C)C)nc2F)n(C)c1=O |r,wD:4.6,2.1,(3.02,4.9,;2.62,3.41,;3.71,2.32,;3.71,.78,;5.25,.78,;5.25,2.32,;6.34,-.31,;6.02,-1.82,;7.35,-2.59,;7.35,-4.13,;6.02,-4.9,;4.69,-4.13,;3.35,-4.9,;2.02,-4.13,;.68,-4.9,;2.02,-2.59,;3.35,-1.82,;4.69,-2.59,;.68,-1.82,;.68,-.28,;-.65,.49,;-1.98,-.28,;-3.32,.49,;-4.65,-.28,;-5.98,.49,;-7.32,-.28,;-8.65,.49,;-9.98,-.28,;-8.65,2.03,;-1.98,-1.82,;-.65,-2.59,;-.65,-4.13,;8.5,-1.56,;9.98,-1.95,;7.87,-.15,;8.64,1.19,)| Show InChI InChI=1S/C26H29F2N5O3/c1-31(2)8-5-9-36-23-7-6-17(25(28)30-23)18-12-19-21(13-20(18)27)29-14-22-24(19)33(26(34)32(22)3)15-10-16(11-15)35-4/h6-7,12-16H,5,8-11H2,1-4H3/t15-,16+ | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human ERG by electrophysiology assay |
J Med Chem 61: 3823-3841 (2018)
Article DOI: 10.1021/acs.jmedchem.7b01896
BindingDB Entry DOI: 10.7270/Q2RB775P |
More data for this
Ligand-Target Pair | |
Serine-protein kinase ATM
(Homo sapiens (Human)) | BDBM245492

(US9428503, 19)Show SMILES CO[C@H]1C[C@H](C1)n1c2c(cnc3cc(F)c(cc23)-c2ccc(OCCCN(C)C)nc2F)n(C)c1=O |r,wD:4.6,2.1,(3.02,4.9,;2.62,3.41,;3.71,2.32,;3.71,.78,;5.25,.78,;5.25,2.32,;6.34,-.31,;6.02,-1.82,;7.35,-2.59,;7.35,-4.13,;6.02,-4.9,;4.69,-4.13,;3.35,-4.9,;2.02,-4.13,;.68,-4.9,;2.02,-2.59,;3.35,-1.82,;4.69,-2.59,;.68,-1.82,;.68,-.28,;-.65,.49,;-1.98,-.28,;-3.32,.49,;-4.65,-.28,;-5.98,.49,;-7.32,-.28,;-8.65,.49,;-9.98,-.28,;-8.65,2.03,;-1.98,-1.82,;-.65,-2.59,;-.65,-4.13,;8.5,-1.56,;9.98,-1.95,;7.87,-.15,;8.64,1.19,)| Show InChI InChI=1S/C26H29F2N5O3/c1-31(2)8-5-9-36-23-7-6-17(25(28)30-23)18-12-19-21(13-20(18)27)29-14-22-24(19)33(26(34)32(22)3)15-10-16(11-15)35-4/h6-7,12-16H,5,8-11H2,1-4H3/t15-,16+ | PDB
UniProtKB/SwissProt
UniProtKB/TrEMBL
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of ATM autophosphorylation at Ser1981 in human HT29 cells preincubated for 1 hr followed by X ray irradiation and measured after 1 hr by H... |
J Med Chem 61: 3823-3841 (2018)
Article DOI: 10.1021/acs.jmedchem.7b01896
BindingDB Entry DOI: 10.7270/Q2RB775P |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase mTOR
(Homo sapiens (Human)) | BDBM245492

(US9428503, 19)Show SMILES CO[C@H]1C[C@H](C1)n1c2c(cnc3cc(F)c(cc23)-c2ccc(OCCCN(C)C)nc2F)n(C)c1=O |r,wD:4.6,2.1,(3.02,4.9,;2.62,3.41,;3.71,2.32,;3.71,.78,;5.25,.78,;5.25,2.32,;6.34,-.31,;6.02,-1.82,;7.35,-2.59,;7.35,-4.13,;6.02,-4.9,;4.69,-4.13,;3.35,-4.9,;2.02,-4.13,;.68,-4.9,;2.02,-2.59,;3.35,-1.82,;4.69,-2.59,;.68,-1.82,;.68,-.28,;-.65,.49,;-1.98,-.28,;-3.32,.49,;-4.65,-.28,;-5.98,.49,;-7.32,-.28,;-8.65,.49,;-9.98,-.28,;-8.65,2.03,;-1.98,-1.82,;-.65,-2.59,;-.65,-4.13,;8.5,-1.56,;9.98,-1.95,;7.87,-.15,;8.64,1.19,)| Show InChI InChI=1S/C26H29F2N5O3/c1-31(2)8-5-9-36-23-7-6-17(25(28)30-23)18-12-19-21(13-20(18)27)29-14-22-24(19)33(26(34)32(22)3)15-10-16(11-15)35-4/h6-7,12-16H,5,8-11H2,1-4H3/t15-,16+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.37E+4 | n/a | n/a | n/a | n/a | n/a | 37 |
AstraZeneca AB
US Patent
| Assay Description
Assay d) MDA-MB-468 cells (human breast adenocarcinoma ##ATCC HTB 132) were seeded at 1500 cells/well in 40 μl of DMEM containing 10% FBS and 1% g... |
US Patent US9428503 (2016)
BindingDB Entry DOI: 10.7270/Q2GH9GVJ |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data






