

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM323490 (1,2-Trans)-2-((3-((R)-3-Amino-1-Hydroxypropyl)Phenoxy)Methyl)Cyclohexyl Acetate::US10188615, Example 173::US10639286, Example 173
SMILES: CC(=O)O[C@@H]1CCCC[C@H]1COc1cccc(c1)[C@H](O)CCN
InChI Key: InChIKey=QVFHDMUQYSVRAM-CGTJXYLNSA-N
Data: 2 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Retinaldehyde-binding protein 1 (Homo sapiens (Human)) | BDBM323490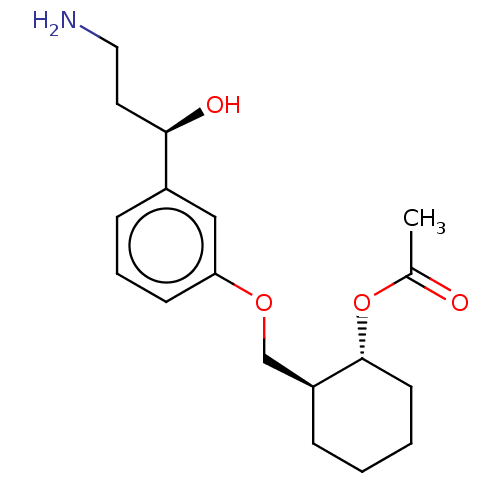 ((1,2-Trans)-2-((3-((R)-3-Amino-1-Hydroxypropyl)Phe...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 55 | n/a | n/a | n/a | n/a | n/a | n/a |
ACUCELA INC. US Patent | Assay Description Isomerase inhibition reactions were performed essentially as described (Stecher et al., J Biol. Chem. 274:8577-85 (1999). | US Patent US10639286 (2020) | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoid isomerohydrolase (Homo sapiens (Human)) | BDBM323490 ((1,2-Trans)-2-((3-((R)-3-Amino-1-Hydroxypropyl)Phe...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 55 | n/a | n/a | n/a | n/a | n/a | n/a |
ACUCELA INC. US Patent | Assay Description Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P... | US Patent US10188615 (2019) BindingDB Entry DOI: 10.7270/Q2NP26HC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||