

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM370517 1-(4-(1-hydroxy-2-(5H-imidazo[5,1-a]isoindol- 5-yl)ethyl)piperidin-1-yl)-2-(pyridin-4- yl)ethanone::US10233190, Example 1437
SMILES: OC(CC1c2ccccc2-c2cncn12)C1CCN(CC1)C(=O)Cc1ccncc1
InChI Key: InChIKey=ADXRHXZIAQWYPA-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Indoleamine 2,3-dioxygenase (Homo sapiens (Human)) | BDBM370517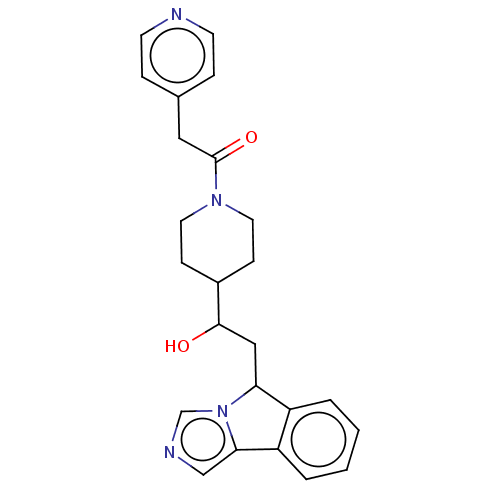 (1-(4-(1-hydroxy-2-(5H-imidazo[5,1-a]isoindol- 5-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | <1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer HealthCare Pharmaceuticals Corporation | Assay Description The IC50 values for each compound were determined by testing the activity of IDO in a mixture containing 50 mM potassium phosphate buffer at pH 6.5; ... | J Med Chem 50: 984-1000 (2007) BindingDB Entry DOI: 10.7270/Q26H4KQM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Indoleamine 2,3-dioxygenase (Homo sapiens (Human)) | BDBM370517 (1-(4-(1-hydroxy-2-(5H-imidazo[5,1-a]isoindol- 5-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 230 | n/a | n/a | n/a | n/a | n/a | n/a |
NewLink Genetics Corporation Curated by ChEMBL | Assay Description Inhibition of purified human IDO1 using L-tryptophan as substrate preincubated for 5 mins followed by substrate addition and measured after 15 mins b... | J Med Chem 62: 6705-6733 (2019) Article DOI: 10.1021/acs.jmedchem.9b00662 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Indoleamine 2,3-dioxygenase (Homo sapiens (Human)) | BDBM370517 (1-(4-(1-hydroxy-2-(5H-imidazo[5,1-a]isoindol- 5-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 1.30E+3 | n/a | n/a | n/a | n/a |
NewLink Genetics Corporation Curated by ChEMBL | Assay Description Inhibition of recombinant human IDO1 expressed in T-REx-293 cells assessed as reduction in kynurenine level measured after 16 hrs | J Med Chem 62: 6705-6733 (2019) Article DOI: 10.1021/acs.jmedchem.9b00662 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2D6 (Homo sapiens (Human)) | BDBM370517 (1-(4-(1-hydroxy-2-(5H-imidazo[5,1-a]isoindol- 5-yl...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
NewLink Genetics Corporation Curated by ChEMBL | Assay Description Inhibition of CYP2D6 (unknown origin) | J Med Chem 62: 6705-6733 (2019) Article DOI: 10.1021/acs.jmedchem.9b00662 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 1A (Homo sapiens (Human)) | BDBM370517 (1-(4-(1-hydroxy-2-(5H-imidazo[5,1-a]isoindol- 5-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
NewLink Genetics Corporation Curated by ChEMBL | Assay Description Inhibition of CYP1A2 (unknown origin) | J Med Chem 62: 6705-6733 (2019) Article DOI: 10.1021/acs.jmedchem.9b00662 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2B6 (Homo sapiens (Human)) | BDBM370517 (1-(4-(1-hydroxy-2-(5H-imidazo[5,1-a]isoindol- 5-yl...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 5.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
NewLink Genetics Corporation Curated by ChEMBL | Assay Description Inhibition of CYP2B6 (unknown origin) | J Med Chem 62: 6705-6733 (2019) Article DOI: 10.1021/acs.jmedchem.9b00662 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 3A4 (Homo sapiens (Human)) | BDBM370517 (1-(4-(1-hydroxy-2-(5H-imidazo[5,1-a]isoindol- 5-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
NewLink Genetics Corporation Curated by ChEMBL | Assay Description Inhibition of CYP3A4 (unknown origin) using midazolam as substrate | J Med Chem 62: 6705-6733 (2019) Article DOI: 10.1021/acs.jmedchem.9b00662 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||