 Found 4 hits for monomerid = 485273
Found 4 hits for monomerid = 485273 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM485273
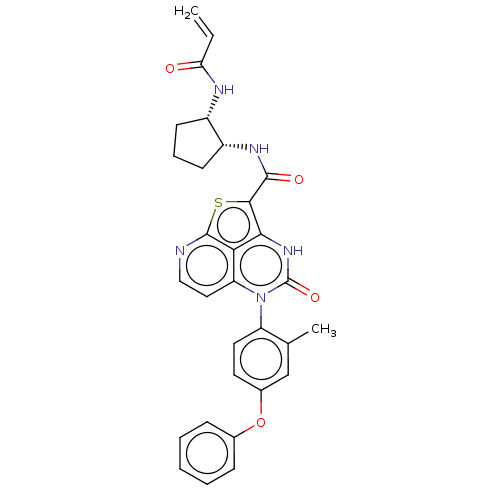
(N-((1R,2S)-2-Acrylamidocyclopentyl)-5-(*S)-(2-meth...)Show SMILES Cc1cc(Oc2ccccc2)ccc1-n1c2ccnc3sc(C(=O)N[C@@H]4CCC[C@@H]4NC(=O)C=C)c([nH]c1=O)c23 |r,wU:25.26,29.32,(-.48,2.9,;-1.82,2.13,;-3.15,2.9,;-4.48,2.13,;-5.82,2.9,;-7.15,2.13,;-8.49,2.9,;-9.82,2.13,;-9.82,.59,;-8.49,-.18,;-7.15,.59,;-4.48,.59,;-3.15,-.18,;-1.82,.59,;-.48,-.18,;-.48,-1.72,;-1.82,-2.49,;-1.82,-4.03,;-.48,-4.8,;.85,-4.03,;2.3,-4.48,;3,-3,;4.54,-3,;5.31,-4.33,;5.31,-1.66,;6.85,-1.66,;7.76,-2.91,;9.22,-2.43,;9.22,-.89,;7.76,-.42,;7.28,1.05,;8.31,2.19,;9.82,1.87,;7.84,3.66,;8.87,4.8,;2.18,-1.72,;2.18,-.18,;.85,.59,;.85,2.13,;.85,-2.49,)| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM485273

(N-((1R,2S)-2-Acrylamidocyclopentyl)-5-(*S)-(2-meth...)Show SMILES Cc1cc(Oc2ccccc2)ccc1-n1c2ccnc3sc(C(=O)N[C@@H]4CCC[C@@H]4NC(=O)C=C)c([nH]c1=O)c23 |r,wU:25.26,29.32,(-.48,2.9,;-1.82,2.13,;-3.15,2.9,;-4.48,2.13,;-5.82,2.9,;-7.15,2.13,;-8.49,2.9,;-9.82,2.13,;-9.82,.59,;-8.49,-.18,;-7.15,.59,;-4.48,.59,;-3.15,-.18,;-1.82,.59,;-.48,-.18,;-.48,-1.72,;-1.82,-2.49,;-1.82,-4.03,;-.48,-4.8,;.85,-4.03,;2.3,-4.48,;3,-3,;4.54,-3,;5.31,-4.33,;5.31,-1.66,;6.85,-1.66,;7.76,-2.91,;9.22,-2.43,;9.22,-.89,;7.76,-.42,;7.28,1.05,;8.31,2.19,;9.82,1.87,;7.84,3.66,;8.87,4.8,;2.18,-1.72,;2.18,-.18,;.85,.59,;.85,2.13,;.85,-2.49,)| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 5.62 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Mus musculus) | BDBM485273

(N-((1R,2S)-2-Acrylamidocyclopentyl)-5-(*S)-(2-meth...)Show SMILES Cc1cc(Oc2ccccc2)ccc1-n1c2ccnc3sc(C(=O)N[C@@H]4CCC[C@@H]4NC(=O)C=C)c([nH]c1=O)c23 |r,wU:25.26,29.32,(-.48,2.9,;-1.82,2.13,;-3.15,2.9,;-4.48,2.13,;-5.82,2.9,;-7.15,2.13,;-8.49,2.9,;-9.82,2.13,;-9.82,.59,;-8.49,-.18,;-7.15,.59,;-4.48,.59,;-3.15,-.18,;-1.82,.59,;-.48,-.18,;-.48,-1.72,;-1.82,-2.49,;-1.82,-4.03,;-.48,-4.8,;.85,-4.03,;2.3,-4.48,;3,-3,;4.54,-3,;5.31,-4.33,;5.31,-1.66,;6.85,-1.66,;7.76,-2.91,;9.22,-2.43,;9.22,-.89,;7.76,-.42,;7.28,1.05,;8.31,2.19,;9.82,1.87,;7.84,3.66,;8.87,4.8,;2.18,-1.72,;2.18,-.18,;.85,.59,;.85,2.13,;.85,-2.49,)| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM485273

(N-((1R,2S)-2-Acrylamidocyclopentyl)-5-(*S)-(2-meth...)Show SMILES Cc1cc(Oc2ccccc2)ccc1-n1c2ccnc3sc(C(=O)N[C@@H]4CCC[C@@H]4NC(=O)C=C)c([nH]c1=O)c23 |r,wU:25.26,29.32,(-.48,2.9,;-1.82,2.13,;-3.15,2.9,;-4.48,2.13,;-5.82,2.9,;-7.15,2.13,;-8.49,2.9,;-9.82,2.13,;-9.82,.59,;-8.49,-.18,;-7.15,.59,;-4.48,.59,;-3.15,-.18,;-1.82,.59,;-.48,-.18,;-.48,-1.72,;-1.82,-2.49,;-1.82,-4.03,;-.48,-4.8,;.85,-4.03,;2.3,-4.48,;3,-3,;4.54,-3,;5.31,-4.33,;5.31,-1.66,;6.85,-1.66,;7.76,-2.91,;9.22,-2.43,;9.22,-.89,;7.76,-.42,;7.28,1.05,;8.31,2.19,;9.82,1.87,;7.84,3.66,;8.87,4.8,;2.18,-1.72,;2.18,-.18,;.85,.59,;.85,2.13,;.85,-2.49,)| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 5.62 | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Pharmaceutica NV
US Patent
| Assay Description
A BTK kinase lanthascreen binding assay monitors compound binding to unphosphorylated-BTK kinase domain (UP-BTK), by competing with a fluorescent lab... |
US Patent US10934310 (2021)
|
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data