

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50070048 (4aR,10bR)-4,10b-Dimethyl-8-thiophen-2-yl-1,4,4a,5,6,10b-hexahydro-2H-benzo[f]quinolin-3-one::CHEMBL341088
SMILES: CN1[C@@H]2CCc3cc(ccc3[C@@]2(C)CCC1=O)-c1cccs1
InChI Key: InChIKey=GUFSVRJDNQUVLC-IEBWSBKVSA-N
Data: 2 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Steroid 5-alpha-reductase (Homo sapiens (Human)) | BDBM50070048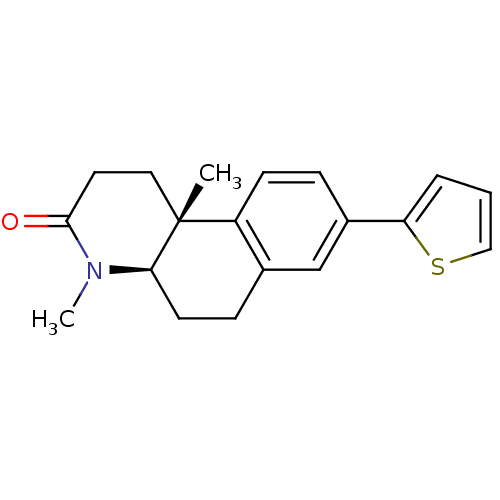 ((4aR,10bR)-4,10b-Dimethyl-8-thiophen-2-yl-1,4,4a,5...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibitory activity against Steroid 5-alpha-reductase type 2 as [3H]-T to [3H]-DHT conversion human prostate nuclear membrane | Bioorg Med Chem Lett 8: 395-8 (1999) BindingDB Entry DOI: 10.7270/Q2CN732C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5α-Reductase 1 (5α-R1) (Homo sapiens (Human)) | BDBM50070048 ((4aR,10bR)-4,10b-Dimethyl-8-thiophen-2-yl-1,4,4a,5...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibitory activity against Steroid 5-alpha-reductase type 1 enzyme based on the conversion of [3H]-T to [3H]-DHT in nuclear membrane preparations fr... | Bioorg Med Chem Lett 8: 395-8 (1999) BindingDB Entry DOI: 10.7270/Q2CN732C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||