

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50076267 (2S,3S)-2,3-Bis-[(E)-3-(3,4-dihydroxy-phenyl)-acryloyloxy]-succinic acid::CHEMBL29660::D-chicoric acid::chicoric acid
SMILES: OC(=O)[C@@H](OC(=O)\C=C\c1ccc(O)c(O)c1)[C@H](OC(=O)\C=C\c1ccc(O)c(O)c1)C(O)=O
InChI Key: InChIKey=YDDGKXBLOXEEMN-QFZCZCNSSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50076267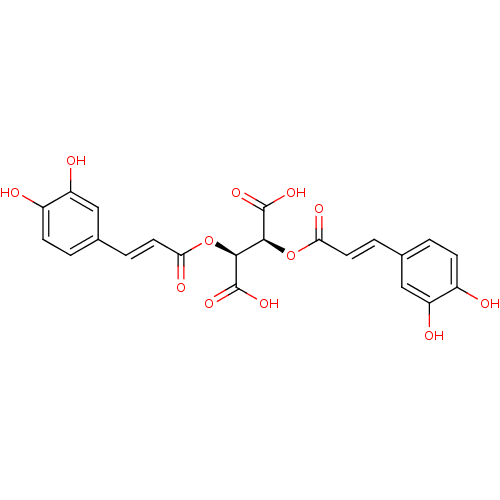 ((2S,3S)-2,3-Bis-[(E)-3-(3,4-dihydroxy-phenyl)-acry...) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Stony Brook University Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A light chain using SNAP-25 as substrate by LC/MS analysis | Bioorg Med Chem 24: 4875-4889 (2016) Article DOI: 10.1016/j.bmc.2016.07.031 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50076267 ((2S,3S)-2,3-Bis-[(E)-3-(3,4-dihydroxy-phenyl)-acry...) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium botulinum full length BoNT/A light chain (1-448) using truncated SNAP 25 (141-206) peptide as substrate by LC/M... | ACS Med Chem Lett 4: 283-287 (2013) Article DOI: 10.1021/ml300428s BindingDB Entry DOI: 10.7270/Q20K29WM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50076267 ((2S,3S)-2,3-Bis-[(E)-3-(3,4-dihydroxy-phenyl)-acry...) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Uncompetitive inhibition of Clostridium botulinum truncated BoNT/A light chain (1-425) using truncated SNAP 25 (141-206) peptide as substrate by LC/M... | ACS Med Chem Lett 4: 283-287 (2013) Article DOI: 10.1021/ml300428s BindingDB Entry DOI: 10.7270/Q20K29WM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50076267 ((2S,3S)-2,3-Bis-[(E)-3-(3,4-dihydroxy-phenyl)-acry...) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 3.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Uncompetitive inhibition of Clostridium botulinum full length BoNT/A light chain (1-448) using truncated SNAP 25 (141-206) peptide as substrate by LC... | ACS Med Chem Lett 4: 283-287 (2013) Article DOI: 10.1021/ml300428s BindingDB Entry DOI: 10.7270/Q20K29WM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50076267 ((2S,3S)-2,3-Bis-[(E)-3-(3,4-dihydroxy-phenyl)-acry...) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 6.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium botulinum truncated BoNT/A light chain (1-425) using truncated SNAP 25 (141-206) peptide as substrate by LC/MS ... | ACS Med Chem Lett 4: 283-287 (2013) Article DOI: 10.1021/ml300428s BindingDB Entry DOI: 10.7270/Q20K29WM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Human immunodeficiency virus type 1 integrase (Human immunodeficiency virus 1) | BDBM50076267 ((2S,3S)-2,3-Bis-[(E)-3-(3,4-dihydroxy-phenyl)-acry...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 500 | n/a | n/a | n/a | n/a | n/a | n/a |
National Cancer Institute Curated by ChEMBL | Assay Description Inhibition of HIV-1 integrase by radiolabeled oligonucleotide-based assay integration | J Med Chem 42: 1401-14 (1999) Article DOI: 10.1021/jm980531m BindingDB Entry DOI: 10.7270/Q2RJ4HPN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Human immunodeficiency virus type 1 integrase (Human immunodeficiency virus 1) | BDBM50076267 ((2S,3S)-2,3-Bis-[(E)-3-(3,4-dihydroxy-phenyl)-acry...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
National Cancer Institute Curated by ChEMBL | Assay Description Compound was evaluated for the inhibitory concentration against HIV-1 integrase by radiolabeled oligonucleotide-based assay 3''-processing | J Med Chem 42: 1401-14 (1999) Article DOI: 10.1021/jm980531m BindingDB Entry DOI: 10.7270/Q2RJ4HPN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50076267 ((2S,3S)-2,3-Bis-[(E)-3-(3,4-dihydroxy-phenyl)-acry...) | PDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lille 1 Curated by ChEMBL | Assay Description Inhibition of HIV1 integrase using U5A/U5B double-stranded DNA as substrate by ELISA | Eur J Med Chem 43: 2268-71 (2008) Article DOI: 10.1016/j.ejmech.2007.12.013 BindingDB Entry DOI: 10.7270/Q2V69NCN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Human immunodeficiency virus type 1 integrase (Human immunodeficiency virus 1) | BDBM50076267 ((2S,3S)-2,3-Bis-[(E)-3-(3,4-dihydroxy-phenyl)-acry...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 70 | n/a | n/a | n/a | n/a | n/a | n/a |
University of California Curated by ChEMBL | Assay Description Inhibitory activity against Human Immunodeficiency Virus Type 1 integrase (HIV-1 IN) in the disintegration assay. | J Med Chem 42: 497-509 (1999) Article DOI: 10.1021/jm9804735 BindingDB Entry DOI: 10.7270/Q2G161J5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||