

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50236333 CHEMBL4092052
SMILES: NC(=O)c1cccc(c1)-c1ccc(OC(=O)NCCCCN2CCN(CC2)c2cccc(Cl)c2Cl)cc1
InChI Key: InChIKey=RMHUUXBXQCLMOK-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| D(3) dopamine receptor (Homo sapiens (Human)) | BDBM50236333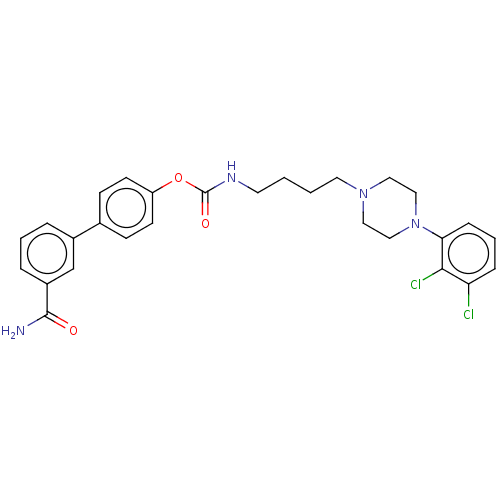 (CHEMBL4092052) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1 | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description Inhibitory concentration against Farnesyltransferase for Farnesylation of H-ras protein InNIH 3T3 cells transformed with activated H-ras | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(2) dopamine receptor (Homo sapiens (Human)) | BDBM50236333 (CHEMBL4092052) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 13 | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description In vitro inhibitory activity against Geranylgeranyl transferase in the geranylgeranylation of H-ras-CAIL protein | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(3) dopamine receptor (Homo sapiens (Human)) | BDBM50236333 (CHEMBL4092052) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | n/a | n/a | n/a | n/a | 1 | n/a | n/a | n/a | n/a | |
TBA | Citation and Details | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50236333 (CHEMBL4092052) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description Inhibition of human FAAH1 expressed in HEK293 cell membrane-enriched lysate using AMC arachidonyl amide as substrate preincubated for 50 mins followe... | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(3) dopamine receptor (Homo sapiens (Human)) | BDBM50236333 (CHEMBL4092052) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 14 | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description Agonist activity at recombinant human CB1 receptor expressed in CHO-K1 cells assessed as increase in cAMP accumulation after 20 mins by HTRF assay | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(3) dopamine receptor (Homo sapiens (Human)) | BDBM50236333 (CHEMBL4092052) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 14 | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description Agonist activity at recombinant human CB1 receptor expressed in CHO-K1 cells assessed as increase in cAMP accumulation after 20 mins by HTRF assay | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||