

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
null
SMILES: C[C@@]1(N)CCCN(CC1)c1nc(N)c(c(n1)C(N)=O)-c1cccc(Cl)c1Cl
InChI Key:
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Tyrosine-protein phosphatase non-receptor type 11 (Homo sapiens (Human)) | BDBM503406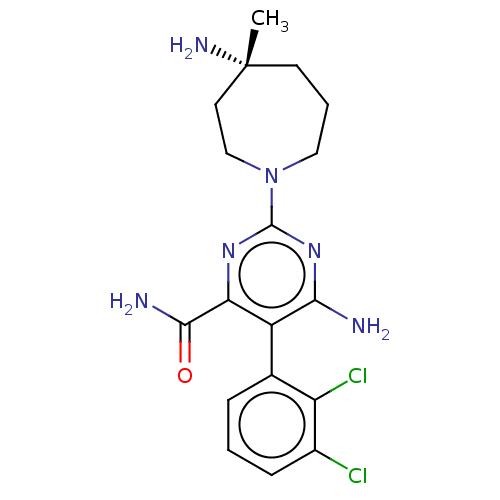 ((5P)-6-amino-2-[(4R)-4-amino-4- methylazepan-1-yl]...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | n/a | n/a | 71 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details BindingDB Entry DOI: 10.7270/Q2JS9VKW | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein phosphatase non-receptor type 11 (Homo sapiens (Human)) | BDBM503406 ((5P)-6-amino-2-[(4R)-4-amino-4- methylazepan-1-yl]...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 71 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing... | Citation and Details BindingDB Entry DOI: 10.7270/Q2930X9N | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||