

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50501640 CHEMBL4102509
SMILES: [Na;v0+].[Na;v0+].[Na;v0+].[#7]-c1ccn(-[#6@@H]-2-[#8]-[#6@H](-[#6]-[#8]P([#8-])(=O)[#8]-[#6@@H](-c3cccc(-[#8]-c4ccccc4)c3)P([#8-])([#8-])=O)-[#6@@H](-[#8])-[#6@H]-2-[#8])c(=O)n1
InChI Key: InChIKey=SHDOSRBONQBOED-GYKYLPKRSA-K
Data: 1 KI
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Beta-galactoside alpha-2,6-sialyltransferase 1 (Homo sapiens (Human)) | BDBM50501640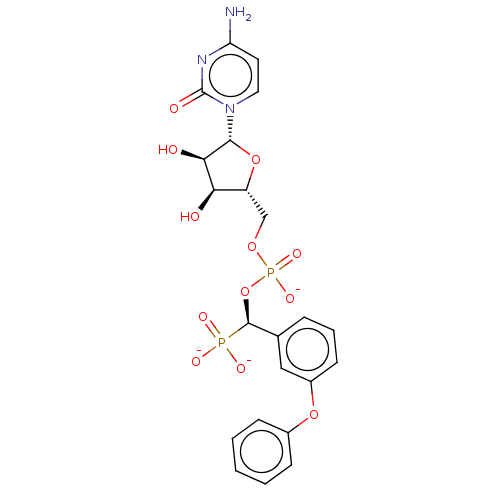 (CHEMBL4102509) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 19 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Peking University Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal His-tagged ST6Gal-1 (44 to 406 residues) using CMP-Neu5Ac as substrate after 20 mins by UV/RP-HPLC method | J Med Chem 60: 2135-2141 (2017) Article DOI: 10.1021/acs.jmedchem.6b01644 BindingDB Entry DOI: 10.7270/Q2XP77Z0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-galactoside alpha-2,6-sialyltransferase 1 (Rattus norvegicus) | BDBM50501640 (CHEMBL4102509) | NCI pathway Reactome pathway KEGG DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem | 70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||