

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50526028 CHEMBL4473888
SMILES: COc1ccccc1-c1c2ccccn2c(=O)n(CCCCN2CCCC(C2)c2c[nH]c3ccccc23)c1=O
InChI Key: InChIKey=JNZLFUGVQULFHJ-UHFFFAOYSA-N
Data: 2 KI
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 5-hydroxytryptamine receptor 1A (Homo sapiens (Human)) | BDBM50526028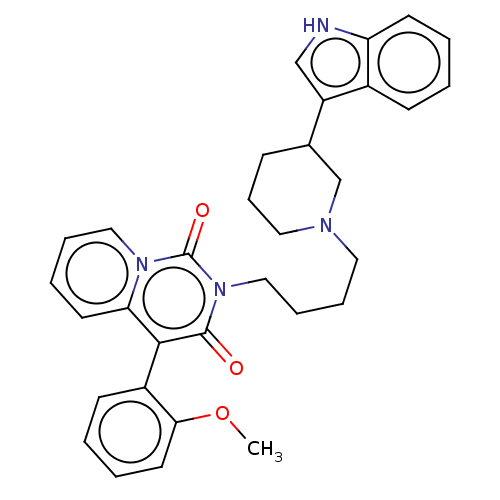 (CHEMBL4473888) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 132 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of Warsaw Curated by ChEMBL | Assay Description Displacement of [3H]-8-OH-DPAT from recombinant human 5HT1A receptor expressed in CHO cell membranes measured after 60 mins by microbeta scintillatio... | Eur J Med Chem 166: 144-158 (2019) Article DOI: 10.1016/j.ejmech.2019.01.031 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sodium-dependent serotonin transporter (Rattus norvegicus (rat)) | BDBM50526028 (CHEMBL4473888) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 411 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of Warsaw Curated by ChEMBL | Assay Description Displacement of [3H]-citalopram from SERT in rat cortex tissue measured after 60 mins by microbeta scintillation counting analysis | Eur J Med Chem 166: 144-158 (2019) Article DOI: 10.1016/j.ejmech.2019.01.031 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||