 Found 3 hits for monomerid = 50544022
Found 3 hits for monomerid = 50544022 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Bile acid receptor FXR
(Homo sapiens (Human)) | BDBM50544022
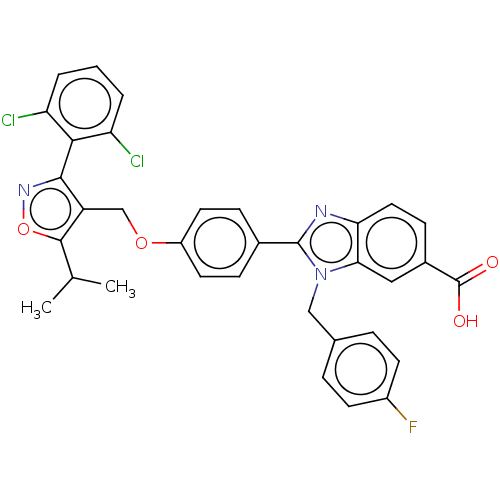
(CHEMBL4644859)Show SMILES CC(C)c1onc(c1COc1ccc(cc1)-c1nc2ccc(cc2n1Cc1ccc(F)cc1)C(O)=O)-c1c(Cl)cccc1Cl |(37.39,-34.35,;38.01,-32.94,;39.55,-32.78,;37.11,-31.7,;35.57,-31.7,;35.09,-30.23,;36.34,-29.32,;37.58,-30.23,;39.05,-29.75,;40.2,-30.78,;41.67,-30.31,;42.81,-31.34,;44.27,-30.87,;44.6,-29.36,;43.44,-28.32,;41.98,-28.81,;46.06,-28.88,;47.3,-29.78,;48.55,-28.87,;50.05,-29.18,;51.08,-28.04,;50.59,-26.57,;49.09,-26.27,;48.07,-27.41,;46.53,-27.41,;45.62,-26.16,;46.24,-24.76,;47.77,-24.61,;48.39,-23.2,;47.49,-21.96,;48.1,-20.56,;45.95,-22.13,;45.34,-23.53,;51.61,-25.42,;53.12,-25.74,;51.13,-23.96,;36.33,-27.78,;37.67,-27,;39,-27.77,;37.66,-25.45,;36.32,-24.69,;34.99,-25.46,;34.99,-27,;33.66,-27.78,)| Show InChI InChI=1S/C34H26Cl2FN3O4/c1-19(2)32-25(31(39-44-32)30-26(35)4-3-5-27(30)36)18-43-24-13-8-21(9-14-24)33-38-28-15-10-22(34(41)42)16-29(28)40(33)17-20-6-11-23(37)12-7-20/h3-16,19H,17-18H2,1-2H3,(H,41,42) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | n/a | n/a | 105 | n/a | n/a | n/a | n/a |
Hiroshima International University
Curated by ChEMBL
| Assay Description
Agonist activity at GST-fused FXR-LBD (unknown origin) incubated with Fluorecein-SRC2-2 coactivator peptide as substrate by TR-FRET assay |
Bioorg Med Chem 28: (2020)
|
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha/Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50544022

(CHEMBL4644859)Show SMILES CC(C)c1onc(c1COc1ccc(cc1)-c1nc2ccc(cc2n1Cc1ccc(F)cc1)C(O)=O)-c1c(Cl)cccc1Cl |(37.39,-34.35,;38.01,-32.94,;39.55,-32.78,;37.11,-31.7,;35.57,-31.7,;35.09,-30.23,;36.34,-29.32,;37.58,-30.23,;39.05,-29.75,;40.2,-30.78,;41.67,-30.31,;42.81,-31.34,;44.27,-30.87,;44.6,-29.36,;43.44,-28.32,;41.98,-28.81,;46.06,-28.88,;47.3,-29.78,;48.55,-28.87,;50.05,-29.18,;51.08,-28.04,;50.59,-26.57,;49.09,-26.27,;48.07,-27.41,;46.53,-27.41,;45.62,-26.16,;46.24,-24.76,;47.77,-24.61,;48.39,-23.2,;47.49,-21.96,;48.1,-20.56,;45.95,-22.13,;45.34,-23.53,;51.61,-25.42,;53.12,-25.74,;51.13,-23.96,;36.33,-27.78,;37.67,-27,;39,-27.77,;37.66,-25.45,;36.32,-24.69,;34.99,-25.46,;34.99,-27,;33.66,-27.78,)| Show InChI InChI=1S/C34H26Cl2FN3O4/c1-19(2)32-25(31(39-44-32)30-26(35)4-3-5-27(30)36)18-43-24-13-8-21(9-14-24)33-38-28-15-10-22(34(41)42)16-29(28)40(33)17-20-6-11-23(37)12-7-20/h3-16,19H,17-18H2,1-2H3,(H,41,42) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 1.14E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Hiroshima International University
Curated by ChEMBL
| Assay Description
Antagonist activity at human VDR transfected in HEK293T cells measured after 24 hrs by luciferase reporter gene assay |
Bioorg Med Chem 28: (2020)
|
More data for this
Ligand-Target Pair | |
Bile acid receptor FXR
(Homo sapiens (Human)) | BDBM50544022

(CHEMBL4644859)Show SMILES CC(C)c1onc(c1COc1ccc(cc1)-c1nc2ccc(cc2n1Cc1ccc(F)cc1)C(O)=O)-c1c(Cl)cccc1Cl |(37.39,-34.35,;38.01,-32.94,;39.55,-32.78,;37.11,-31.7,;35.57,-31.7,;35.09,-30.23,;36.34,-29.32,;37.58,-30.23,;39.05,-29.75,;40.2,-30.78,;41.67,-30.31,;42.81,-31.34,;44.27,-30.87,;44.6,-29.36,;43.44,-28.32,;41.98,-28.81,;46.06,-28.88,;47.3,-29.78,;48.55,-28.87,;50.05,-29.18,;51.08,-28.04,;50.59,-26.57,;49.09,-26.27,;48.07,-27.41,;46.53,-27.41,;45.62,-26.16,;46.24,-24.76,;47.77,-24.61,;48.39,-23.2,;47.49,-21.96,;48.1,-20.56,;45.95,-22.13,;45.34,-23.53,;51.61,-25.42,;53.12,-25.74,;51.13,-23.96,;36.33,-27.78,;37.67,-27,;39,-27.77,;37.66,-25.45,;36.32,-24.69,;34.99,-25.46,;34.99,-27,;33.66,-27.78,)| Show InChI InChI=1S/C34H26Cl2FN3O4/c1-19(2)32-25(31(39-44-32)30-26(35)4-3-5-27(30)36)18-43-24-13-8-21(9-14-24)33-38-28-15-10-22(34(41)42)16-29(28)40(33)17-20-6-11-23(37)12-7-20/h3-16,19H,17-18H2,1-2H3,(H,41,42) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | n/a | n/a | 3.30 | n/a | n/a | n/a | n/a |
Hiroshima International University
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR expressed in human HuH7 cells by luciferase reporter gene assay |
Bioorg Med Chem 28: (2020)
|
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data


